‘Wondrous’ Drug to Treat Aggressive Leukaemia Gets UK Approval
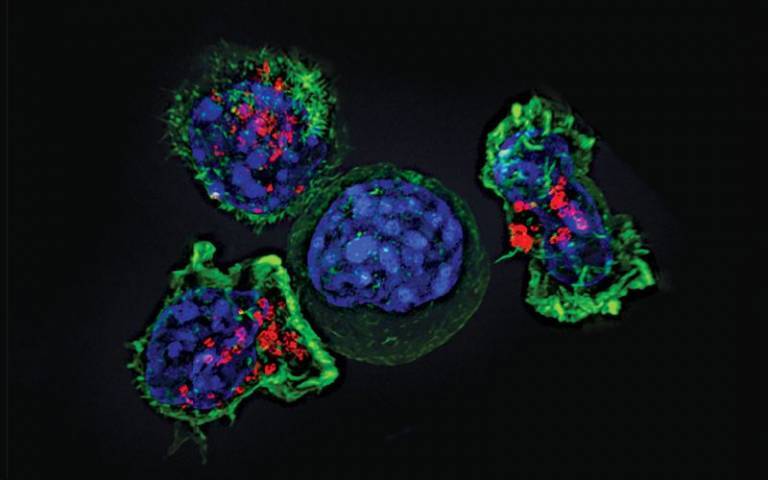
Adult patients with an aggressive form of leukaemia will be able to receive a breakthrough immunotherapy, which was invented by University College London researchers, on the NHS within weeks following approval for use by the UK’s National Institute for Health and Care Excellence (NICE).
The CAR T-cell therapy – known as ‘obe-cel’ and marketed as Aucatzyl – involves taking a patient’s immune cells and reprogramming them in a lab to identify and target their cancer, before returning them to the body as ‘living medicine’.
Obe-cel is a second-generation CAR T cell therapy invented by scientists from the UCL Cancer Institute, led by Dr Martin Pule, and has delivered promising results in treating patients with acute lymphoblastic leukaemia (ALL), an aggressive blood cancer.
The therapy has reduced immune toxicity and persists for longer in blood cancer patients, overcoming two common limitations of earlier CAR T cell therapies. Aucatzyl was taken through clinical trials and is manufactured by UCL spinout business Autolus, which was set up with the help of UCL Business, the commercialisation company of UCL.
The development of CAR T cell therapy has had long-standing support from the National Institute for Health and Care Research (NIHR) UCLH Biomedical Research Centre (BRC).
NHS England today announced that the personalised therapy would be available on the NHS within weeks through specialist centres.
Dr Claire Roddie, one of the team who developed the treatment from UCL Cancer Institute and UCLH consultant haematologist, said: “I am delighted to hear of NICE’s decision. Many more patients now stand to benefit from CAR-T cell therapy on the NHS.
“We have been working on proving the safety and efficacy of this drug since 2017 and it has brought together clinical and research teams from UCL and UCLH, with support from government and arm’s-length bodies like the NIHR and the BRC as well as the pharmaceutical industry.
“The many, many people involved in this work can feel immensely proud of this achievement which will help save the lives of many more patients.”
Eligible patients will receive two doses of CAR-T therapy intravenously, ten days apart, with the treatment being delivered at specialist CAR-T centres across the country.
The treatment will be available to people aged 26 and over with B-cell acute lymphoblastic leukaemia which has returned or not responded to previous treatment.
It is estimated that it could be administered to around 50 patients each year in England.
In a clinical trial, 77% of patients saw their cancer enter remission after treatment with obe-cel, with half of those showing no signs of detectable cancer after three and a half years.
The treatment – which has been researched, developed and manufactured in the UK – was also found to have lower toxicity and was less likely to cause serious side effects than other CAR (chimeric antigen receptor) T-cell therapies.
Dr Anne Lane, UCL Business CEO, said: “This cutting-edge personalised immunotherapy has been on a 10-year journey starting with research by clinical academics in UCL’s Cancer Institute who, with the support of UCL Business, established Autolus, a spinout company dedicated to developing, trialling and bringing AUCATZYL® to market. That journey has required vision, tenacity and over £800m. Today that has hugely paid off and will benefit people across the UK. It’s an inspiring demonstration of what can be achieved when university academics, NHS hospitals and investors work together.”
Professor Peter Johnson, NHS National Clinical Director for Cancer, said: “This cutting-edge therapy has shown real promise in trials and could give patients with this aggressive form of leukaemia a chance to live free from cancer for longer – and, for some, it could offer the hope of a cure.
“This ‘living medicine’ boosts a patient’s own immune system and then guides T-cells towards the cancer to kill it – it is fantastic to have another pioneering option available on the NHS, adding to our range of CAR-T therapies which are helping people with blood cancers live longer, healthier lives.”
Harry, a 19-year-old student from Harrogate, was treated with obe-cel for B-cell ALL as part of a clinical trial in 2024. He said: “I feel so lucky to have had access to such a wondrous treatment. Not only did it work better than my doctors thought it would, it worked without many of the horrible side effects you can get from other treatments.
“I think it’s brilliant obe-cel is now available on the NHS for people over the age of 26. The biggest thing it offers is hope. When you’re facing a situation like mine, hope is the most valuable thing you can have.”
Health Minister Ashley Dalton said: “This pioneering treatment is excellent news for patients and their families, demonstrating how the NHS is at the forefront of medical innovation.
“Our 10 Year Health Plan is about harnessing our world-leading life sciences sector to deliver treatments like this – innovative therapies that save lives.
“By supporting new treatments with fewer side effects and shorter hospital stays, we’re building an NHS fit for the future whilst cementing the UK’s position as a global leader in medical research.”
Fiona Bride, interim Chief Commercial Officer and Director of Medicines Value & Access at NHS England, said: “This is a success story that’s made in Britain, and shows how collaboratively we can bring to life the ambition of the 10 Year Health Plan, showcasing how the UK’s competitive edge in life sciences can translate to better outcomes and treatments for NHS patients.
“The journey of obe-cel from scientific research in a UK university to a safe, clinically and cost-effective treatment set to be delivered through the NHS specialist CAR-T network is a remarkable one and I am grateful to colleagues who have played their part along the way.”
Acute lymphoblastic leukaemia is an aggressive cancer in the blood and bone marrow, with around 800 people being diagnosed in the UK every year, around half of which are in adults.
Data shows patients with aggressive forms of the cancer receiving chemotherapy, the current routine standard of care, live for just 10 months on average after treatment.
The therapy will be fast tracked to patients more quickly than the standard 90-day implementation period thanks to interim funding from the NHS’s Cancer Drugs Fund.
Source: University College London