Honey from Australian Wildflowers has Potent Power to Kill Bacteria

Kenya Fernandes, University of Sydney
Before antibiotics and antiseptics, healers across ancient Egypt, Greece, and China reached for honey to treat wounds. Archaeological evidence shows humans have been harvesting and collecting honey for thousands of years – and for much of that time, we understood it to be more than just food.
Today, honey sits in most kitchen cupboards as a perfectly ordinary pantry staple. But honey has never entirely shed its medicinal reputation. And modern research shows us why: it possesses genuine antimicrobial properties, capable of killing or inhibiting a wide range of bacteria, including drug-resistant strains.
This matters now more than ever. Antimicrobial resistance – where bacteria evolve to survive drugs designed to kill them – is one of the defining public health crises of our time. Infections caused by these resistant microbes are becoming harder and more expensive to treat, creating an urgent need for alternative therapies.
Our new study, published in the journal MicrobiologyOpen, shows honeys from Australia’s native flora might be a big part of the solution.
What did we do?
We analysed 56 honey samples collected from more than 35 apiaries across New South Wales. Many samples came from landscapes recovering from the 2019–2020 bushfires. Most were derived from native Australian plants such as eucalyptus, leptospermum and melaleuca.
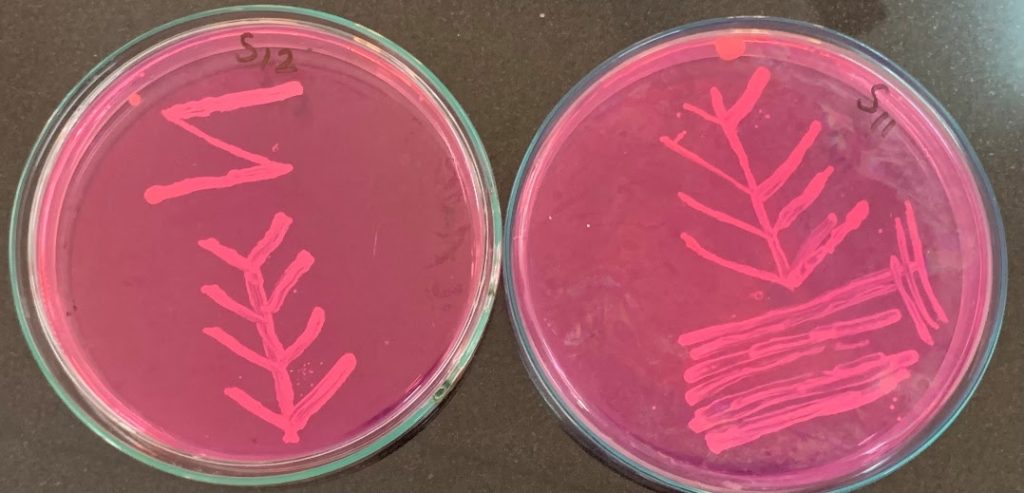
We tested the honeys against two common bacterial pathogens: Staphylococcus aureus (golden staph) and E. coli – both among the six leading causes of deaths associated with antibiotic resistance. For each sample we measured the minimum concentration needed to stop bacterial growth. The lower the concentration, the more potent the honey.
We also carried out comprehensive chemical profiling, measuring sugars, organic acids, amino acids, enzymes and a wide range of plant-derived compounds. Statistical and machine-learning analyses helped us identify which chemical features best explained antibacterial strength.
What did we find?
More than three-quarters of the honey samples stopped bacterial growth even when the honeys were diluted to 10% or less. This places Australian native flora honeys alongside some of the world’s most potent varieties.
The most striking factor was floral diversity.
Honeys from mixed floral sources – where bees foraged across multiple native plant species rather than a single species – were consistently the most antimicrobial.
This potency wasn’t due to any single compound but to a chemically rich combination.
Multiple bioactive factors – substances that have a measurable effect on living cells or tissues – worked together to inhibit bacteria. These included naturally produced hydrogen peroxide, plant-derived phenolic compounds (naturally occurring chemicals that plants produce as part of their own defence systems), and antioxidants.
When bacteria encounter honey, this combination acts on several fronts at once. The low moisture content draws water out of bacterial cells, while the acidity disrupts their metabolism. Hydrogen peroxide damages their cellular structures, and phenolic and antioxidant compounds interfere with their ability to function and reproduce.
The strength of mixed floral honeys may also reflect the health of the bees themselves.
Access to diverse forage keeps colonies well nourished. And healthier bees produce more biologically active honey as their enzymes help integrate and activate the plant compounds into a complex antimicrobial mixture.
What does this mean for antimicrobial resistance?
Honey won’t replace antibiotics for serious or systemic infections.
But for topical applications – chronic wounds, burns, or surgical site infections – it is a genuinely promising option. Because honey attacks bacteria through multiple simultaneous mechanisms, resistance is far less likely to emerge than with single-target drugs. Our team is now exploring these applications in more detail.
Australia is particularly well-placed to lead in bioactive honey production. Around 70% of Australian honey comes from native plants. These plants are found not only in forests but also across farmland, regional landscapes, and urban green spaces.
Our findings show that prioritising floral diversity over monoculture isn’t just good for ecosystems – it produces more potent honey. With the beekeeping industry under serious pressure from bushfires, floods, and now the varroa mite, protecting and restoring florally-rich landscapes is critical: for bee health, for industry resilience, and for expanding our natural antimicrobial toolkit.
In the meantime, the next jar of Australian honey you buy may just be doing more good than you realise.
Kenya Fernandes, Research Fellow, Faculty of Science, University of Sydney
This article is republished from The Conversation under a Creative Commons license. Read the original article.