Head Impacts Associated with Altered Gut Microbiome in Football Players
Non-concussive head impacts correlated with changes to the gut microbiome on following days, in pilot study tracking six US collegiate football players over one season

Non-concussive head impacts – hits to the head that don’t cause clinically detectable symptoms – are correlated with subsequent changes to the gut microbiome in a small sample of US collegiate football players, according to a new study published May 6, 2026, in the open-access journal PLOS One by Ahmet Ay and Kenneth Douglas Belanger of Colgate University, USA, and colleagues.
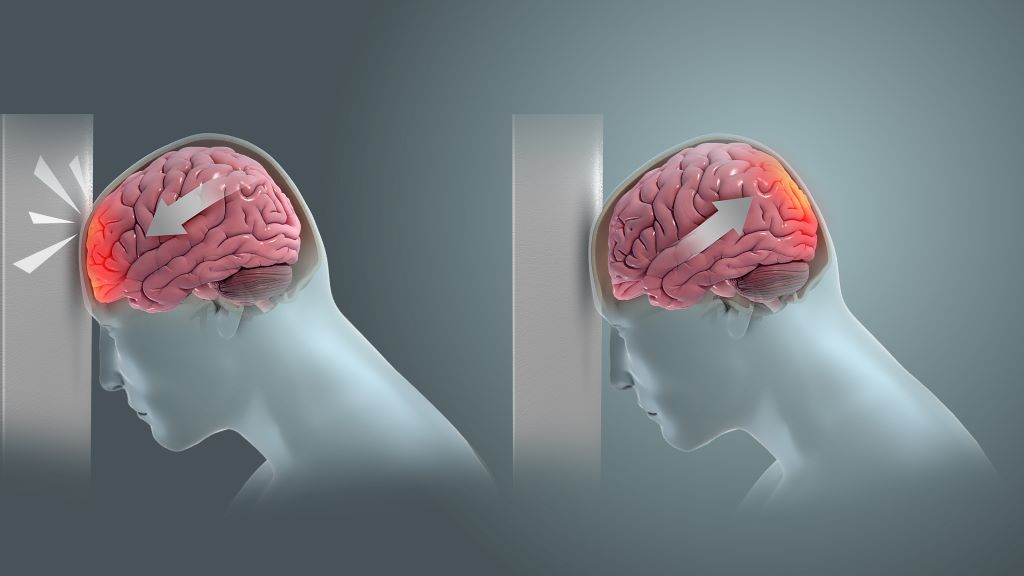
Non-concussive head impacts are common in American football, with players experiencing between 100 and 1000 across a season. While research has shown that full concussions can disrupt the gut microbiome – which regulates inflammation and the neuroimmune system – whether sub-concussive hits might produce similar effects had not been investigated.
In the new study, researchers tracked six NCAA Division I American football players across a competition season, beginning during preseason training. Their on-field activity profiles were monitored using GPS units and head impacts were tracked using a helmet-based sensor system; 226 faecal samples were analysed for their microbiome composition; and participants completed lifestyle questionnaires after each sample collection.
The researchers found that microbial diversity changed within two to three days after a substantial head impact. Specifically, certain bacteria – including the order Coriobacteriales, the family Prevotellaceae, and the genus Prevotella – tended to decrease in abundance while the genus Ruminococcus increased. In previous studies, these changes have correlated with brain injury and inflammation.
The athletes’ gut microbiomes also changed significantly over the course of the season, with mathematical modelling suggesting that the cumulative effects of non-concussive head impacts was likely associated with this shift, even after accounting for 15 potentially confounding factors including diet changes, exercise intensity, sleep, and stress.
The study is limited by its small sample size and lack of a control group, with its design meaning findings could only establish correlation but not causation. However, the authors conclude that even sub-symptomatic head impacts might affect the gut microbiome, both in the immediate aftermath of injury and over a longer time course in athletes who experience multiple impacts.
Ken Belanger adds: “As far as we are aware, this is the first study to examine connections between head impacts and the composition of the gut microbiome – the complex community of bacteria and other organisms within the digestive system.”
“Our results provide evidence that even head impacts that do not result in a concussion or other reported symptoms may influence the microbes present within the gut, both in the short- and longer-term. Determining what causes these changes and whether they have a positive or negative influence on recovery from head injury will require further investigation.”
“Our research highlights the importance of thinking integratively about the interactions between the gut and the brain. We are only beginning to scratch the surface in our understanding of how these complex organs and organ systems communicate with and affect each other.”
Aziz Zafar adds: “After having only heard of the complicated interplay between neuronal inflammation and the gut microbiome, I found it to be such an exciting scientific experience to explore that interplay in the context of head impacts.”
Zachary Pelland adds: “It has been an amazing privilege to work so deeply on a personally and scientifically meaningful project which could not have happened without immeasurable support across academic departments, athletics, administration, and alumni at Colgate University.”
Provided by PLOS