Novel Research Reveals the Active Role that Skin Cells Play in Rabies Infection
New findings identify keratinocytes as replication hubs and immune responders, contributing to the risk of rabies infection from superficial scratches or minor bites

This normal human skin cell was treated with a growth factor that triggered the formation of specialised protein structures that enable the cell to move. We depend on cell movement for such basic functions as wound healing and launching an immune response. Credit: Torsten Wittmann, University of California, San Francisco
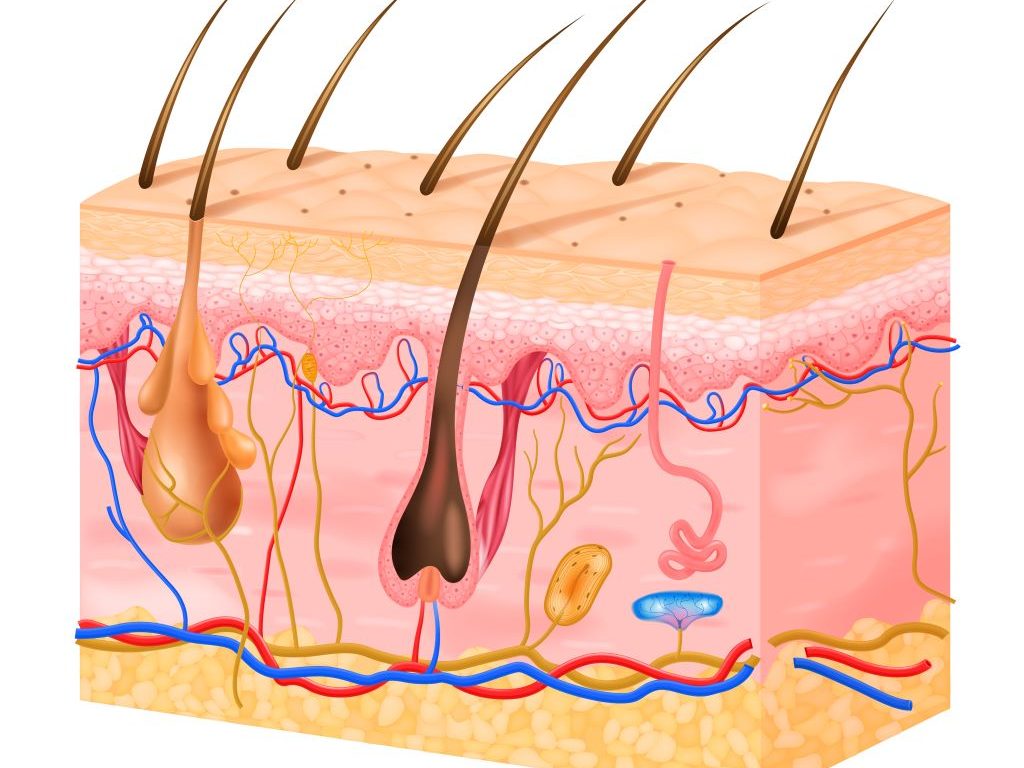
While it was previously thought that keratinocytes (skin cells) were only passive conductors that allow the rabies virus to pass through, novel research reveals that these cells play a much more active role. The findings of a new study in the Journal of Investigative Dermatology (JID), published by Elsevier, provide direct evidence that keratinocytes can support viral replication and transmit the rabies virus to neurons. The investigators offer a mechanistic explanation for how superficial skin exposures from scratches or minor bites by dogs and bats can lead to neuroinvasion, contributing to the risk of infection.
Rabies is a fatal zoonotic infection caused by rabies virus (RABV), responsible for at least 59 000 human deaths per year. The virus is transmitted through the saliva of infected animals. While most cases are caused by dog bites, superficial exposures such as bat bites or scratches can also lead to infection, although the underlying mechanisms remain poorly understood.
“In our previous work, we discovered that keratinocytes – cells that form the epidermis, the outermost layer of the skin – were infected at the site of entry of the rabies virus, both in natural and experimental infections. This was unexpected, as rabies pathogenesis has traditionally focused on muscle cells and motor neurons,” explains lead investigator Corine H. Geurts van Kessel, MD, PhD, Department of Viroscience, Erasmus Medical Centre, Rotterdam, The Netherlands. “Given the strategic position of keratinocytes at the skin barrier and their close proximity to sensory nerve endings, we wanted to understand whether these cells are simply bystanders or active participants in early rabies infection and neuroinvasion.”
The investigators used primary human keratinocyte cultures to investigate susceptibility to rabies virus infection and characterise the resulting antiviral immune responses. Three viral strains were tested: a vaccine strain and two wild-type (“street”) strains derived from fatal human cases associated with bat and dog exposures. The dog-associated strain caused only minimal infection and limited keratinocyte immune activation, whereas the other two strains infected keratinocytes more readily and triggered a pronounced antiviral response.
To simulate the close contact between keratinocytes and intra-epidermal nerve endings, a co-culture model of keratinocytes and neurons was developed. In this model, virus produced in infected keratinocytes was successfully transmitted to adjacent neurons, giving the virus a direct route into the nervous system. Once the virus has established infection in the central nervous system, it is almost inevitably fatal.
“Our study demonstrates that the skin might play a more important role in rabies infection than previously recognised. We were particularly surprised by the strong antiviral response mounted by keratinocytes to the bat-related rabies virus strain,“ notes co-investigator Keshia Kroh, PhD candidate, Department of Viroscience, Erasmus Medical Centre, Rotterdam, The Netherlands. “Wild-type rabies viruses are known for their immunosuppressive capacities, and we expected an immune evasive effect in keratinocytes. Instead, we observed the opposite. This raises new questions about how keratinocyte-derived immune responses influence overall disease progression in rabies and other viral infections of the skin.”
This in vitro co-culture model is the first to study rabies virus entry to the nervous system across a cell barrier. Future in-depth studies should be performed to provide mechanistic insight into the differential strain tropism, the interactions of infected keratinocytes with immune cells, and the mechanisms of neuroinvasion from superficial skin contact.
According to the World Health Organization (WHO), any transdermal exposure (including small scratches or abrasions) should be assessed as a potential rabies risk and managed appropriately based on exposure category and clinical context.
“Our study provides a biological rationale for these recommendations,” says co-investigator Carmen W.E. Embregts, PhD, Department of Viroscience, Erasmus Medical Centre, Rotterdam, The Netherlands. “At the same time, it is important to emphasise that the risk of rabies virus infection via superficial exposures depends on multiple factors, including the nature of the exposure and the epidemiological setting. Rather than causing alarm, our findings support informed decision-making. Awareness that superficial skin exposures can represent a route of neuroinvasion helps ensure that potential risks are recognised and evaluated appropriately, while treatment decisions remain guided by established public health criteria.”
“The data in this study support the increasingly recognised concept that cells in the skin are in snug communication with the nervous system. That a scratch or bite is needed for the transmission of rabies is further evidence of the importance of an intact skin barrier in health,” observes JID Associate Editor Ethan Lerner, MD, PhD, Associate Professor of Dermatology, Harvard Medical School, and Massachusetts General Hospital, Boston, MA, USA.
Source: Elsevier