New Drug Doubles One-year Survival in Pancreatic Cancer Trial
Phase 2 randomised trial shows 38% reduction in risk of death with the drug when combined with chemotherapy
Pancreatic cancer is one of the deadliest cancers and among the hardest to treat, with most patients surviving less than a year after diagnosis. But a new drug developed at Northwestern University may soon help patients live longer.
In a randomised phase 2 clinical trial, patients who received the experimental drug elraglusib, alongside standard chemotherapy, were twice as likely to be alive after one year of treatment, compared to those receiving chemotherapy alone. The drug also reduced the risk of death by 38%.
The study is one of only a few successful randomised trials in the last decade to show a survival benefit that would be applicable to a broad population of pancreatic cancer patients, according to the authors.
The study, which was led by Northwestern Medicine, was published in Nature Medicine.
“Pancreatic cancer remains one of the most challenging solid tumours to treat, but these findings provide cautious optimism for patients,” said study lead author Dr Devalingam Mahalingam, professor of medicine in the division of Hematology and Oncology at Northwestern University Feinberg School of Medicine.
“While these results will need to be confirmed in phase 3 trials, observing survival benefit in such a difficult-to-treat cancer is encouraging. Given the novel mechanism of this drug, these findings raise the possibility that it could have broader application across other tumour types,” added Mahalingam, who also is the associate director of clinical research at Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
How the trial was conducted
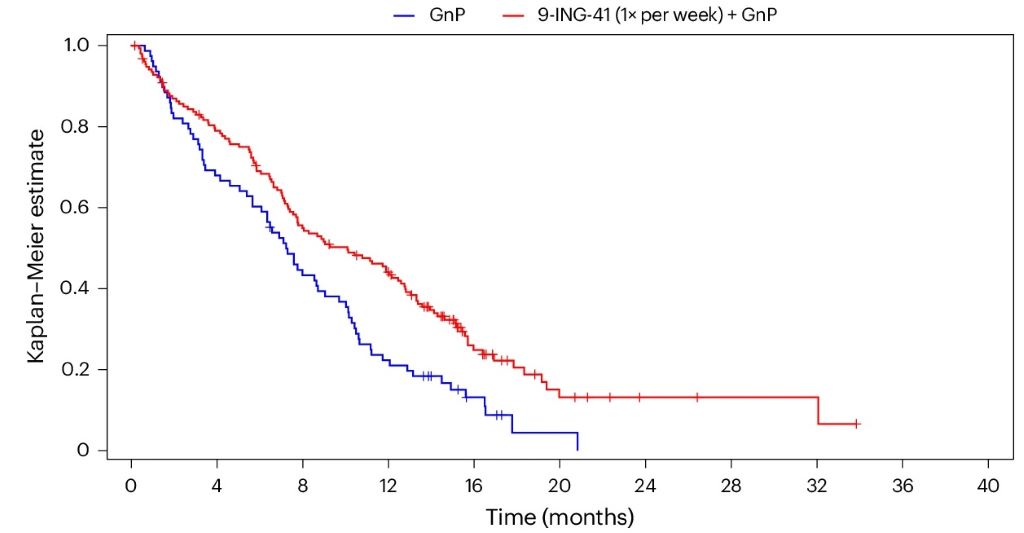
The phase 2 trial enrolled 233 patients with metastatic pancreatic cancer across 60 sites in six countries in North America and Europe. Patients were randomly assigned to receive either standard chemotherapy or the same chemotherapy combined with elraglusib.
Those who received elraglusib lived a median of 10.1 months, compared to 7.2 months for those who only received chemotherapy. While that three-month difference may appear modest, it’s partly because the trial included patients whose cancer progressed too quickly to benefit from treatment.
Among patients who benefited from the drug, the impact was pronounced. Twice as many patients who received elraglusib were alive at one year (44% vs 22%) and about 13% of patients in the drug group were alive at two years, compared to none in the chemotherapy group.
Side effects were generally consistent with chemotherapy but slightly more common in the elraglusib group. The most frequent included low white blood cell counts, fatigue and temporary vision changes, which were reversible. Overall, the safety profile of the drug was considered manageable, the authors said in the study.
How elraglusib works
Elraglusib was developed nearly 15 years ago inside Northwestern University labs. It targets a protein known as GSK-3 beta, which plays a role in tumour growth and suppression of the immune system.
Unlike traditional chemotherapy, which aims to kill cancer cells, elraglusib seems to act on the tumour microenvironment – the mix of cancer cells, immune cells and surrounding tissue that can either support or weaken tumours.
Pancreatic tumours are hard to treat in part because of their microenvironment, which is particularly adept at suppressing immune response. In the study, patients who received elraglusib showed increases in cancer-fighting cells within their tumours, offering early evidence that the drug may help re-engage the immune system.
In addition, certain immune-related markers in the blood at the start of the trial were associated with longer survival among patients who received the drug. While these findings are preliminary, they suggest that elraglusib may be particularly effective in certain patients whose immune systems are already primed to respond.
Mahalingam and his colleagues are exploring a larger confirmatory phase 3 trial as funding and partnership allow. They are also interested in studying the drug in combination with other novel therapies to determine if broader clinical benefit can be achieved.
Source: Northwestern University