Childhood Leukaemia Aggressiveness Depends on Timing of Genetic Mutation
A team of researchers at the Icahn School of Medicine at Mount Sinai has uncovered why children with the same leukaemia-causing gene mutation can have dramatically different outcomes: it depends on when in development the mutation first occurs.
The study, led by Elvin Wagenblast, PhD, Assistant Professor of Oncological Sciences, and Pediatrics, at the Icahn School of Medicine at Mount Sinai, was published in Cancer Discovery. It shows that leukemia beginning before birth is often more aggressive, grows faster, and is harder to treat. This adds a missing dimension to precision medicine for childhood leukaemia.
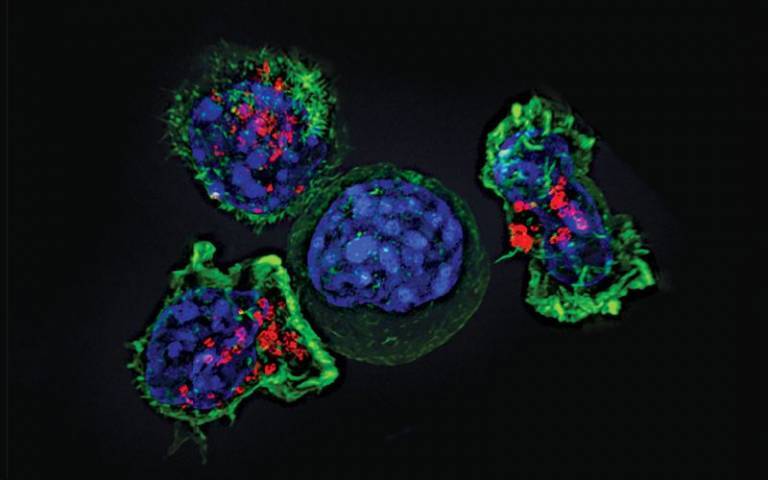
Dr. Wagenblast and his team at the Wagenblast Lab set out to answer a central question about how a normal blood stem cell can become cancerous. They applied cutting-edge CRISPR/Cas9 genome-editing approaches in human primary blood stem cells to model different developmental stages of acute myeloid leukaemia, one of the most aggressive types of blood cancer.
Using CRISPR technology, the team induced the NUP98::NSD1 fusion oncoprotein, a cancer-promoting protein created when two genes abnormally fuse, into human blood stem cells from multiple developmental stages, ranging from prenatal to postnatal, adolescence, and adulthood. This approach created the first humanised experimental model that tracks how the same mutation behaves differently depending on when in life it arises.
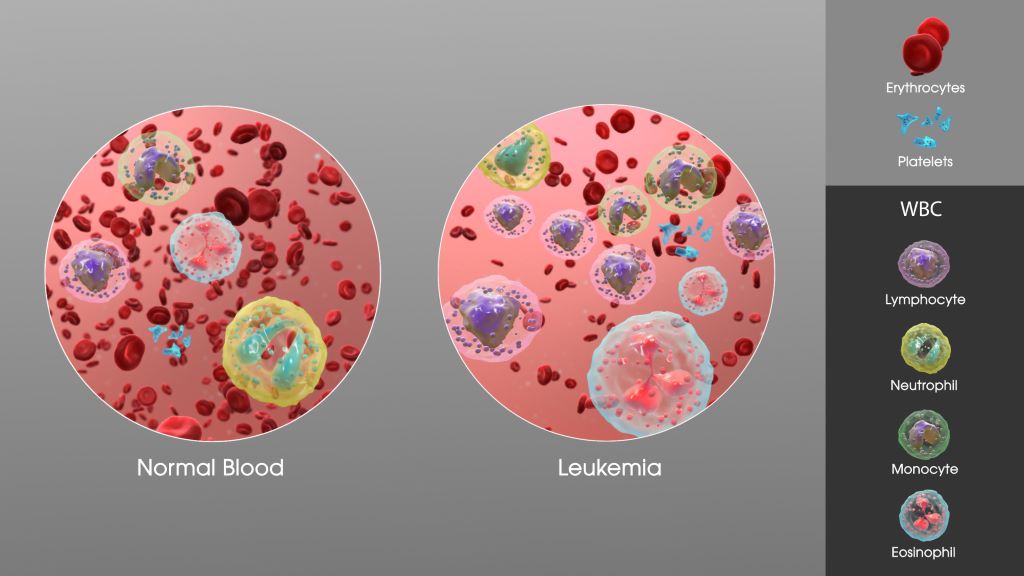
The results were striking: stem cells produced during prenatal development transformed easily into leukaemia, creating a highly aggressive and more primitive form of leukaemia. Stem cells produced postnatally became increasingly resistant to transformation and required additional mutations to become cancerous. Prenatal-origin leukaemia stem cells, which are abnormal blood stem cells that arise before birth and can cause certain childhood leukaemias, were more dormant (quiescent) and relied heavily on certain energy sources specific to the cancer state, which were not seen in the leukaemias that originated later in life. Although these prenatal leukaemia stem cells were more dormant, this quiescent state makes them harder to eliminate with standard treatments, helping explain why prenatal-origin leukaemias behave more aggressively, despite identical genetics.
By analysing single-cell gene expression data from their models, the investigators identified a prenatal gene signature that predicts whether a child’s leukaemia likely began before birth. In patients, this signature strongly correlated with significantly worse clinical outcomes.
“This work tells us that age matters at the cellular level,” said Dr Wagenblast. “The same mutation behaves very differently depending on when it happens. Understanding this gives us a new way to identify the highest-risk patients and to tailor therapies that go beyond standard genetic classifications.”
The team tested therapies against the most aggressive leukaemia stem cells and discovered that these cells were especially vulnerable to venetoclax, a Food and Drug Administration-approved drug already used in the clinic. Venetoclax-based combinations, including with standard chemotherapy, significantly reduced aggressiveness in the experimental models.
“These findings give clinicians mechanistic support to use venetoclax combinations in NUP98-rearranged acute myeloid leukaemia, particularly in younger patients whose disease likely started before birth,” said Dr Wagenblast.
Understanding when leukaemia begins may help doctors choose more effective therapies earlier, reducing trial-and-error approaches and preventing resistance and relapse later on.
Conceptually, the study shifts how scientists understand childhood cancer. The developmental timing of the first mutation is not a minor detail. It fundamentally shapes disease biology, treatment resistance, and relapse risk.
The research opens the door to new diagnostic tools that can identify prenatal-origin leukaemias, venetoclax-based combination therapies that more precisely target vulnerable leukaemia stem cells, and clinical trials that incorporate developmental timing into risk assessment.
Next, the team plans to develop therapies that more directly target the metabolic program unique to prenatal-origin leukaemias, with the goal of selectively eliminating leukaemia stem cells while sparing healthy blood stem cells.
Source: Mount Sinai