Targeted Radiation During Surgery Reduces Pancreatic Cancer Recurrence
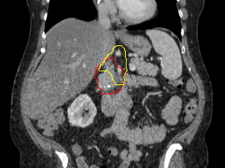
Using targeted radiation during surgery – referred to as intraoperative radiation – to eliminate pancreatic cancer cells that have spread to areas around the pancreas, investigators at Johns Hopkins have been able to reduce the recurrence rate around the pancreas to 5%. This is believed to be the lowest ever reported for this population of patients, according to a preliminary study by the team from the Johns Hopkins Kimmel Cancer Center.
The study was presented at the American Society for Radiation Oncology annual meeting in September 2025.
The study enrolled 20 patients with borderline resectable or locally advanced pancreatic cancer. Patients received presurgical chemotherapy and radiation targeted to shrink the tumours away from the blood vessels. Then, during surgery to remove their tumours, patients received another dose of precisely targeted radiation using a robotic device that carries small radioactive beads inserted through catheters. The device enabled the team to pinpoint a triangular area near the pancreas, where recurrences commonly occur. Only one of the 20 patients experienced a recurrence around the pancreas at the 24-month mark – a major achievement for a cancer that, until recently, had lagged behind other cancers in treatment success.
By the time most pancreatic cancers are diagnosed, the tumours have spread to affect important blood vessels around the pancreas. Historically, patients with pancreatic cancers whose blood vessels were affected could not undergo surgical removal of their tumours. But in the past decade, clinicians at the Johns Hopkins Kimmel Cancer Center’s Skip Viragh Center for Pancreas Cancer Clinical Research and Patient Care have pioneered new approaches that use chemotherapy and radiation to shrink the tumours away from blood vessels, enabling more patients to undergo surgical removal of their tumours.
However, many of these patients continued to experience tumor recurrences, and Amol Narang, M.D., associate professor of radiation oncology and molecular radiation sciences, and his colleagues sought to determine why.
The team learned that the pancreatic cancer cells were spreading along nerves near the pancreas to a fatty, nerve-dense triangular area just above the pancreas, which Narang calls the “Baltimore triangle.” When he and his colleagues started targeting the Baltimore triangle with radiation before surgery to kill these stray cancer cells, pancreatic cancer recurrence rates in their patients dropped from 47% to 12% at two years post-surgery. Yet, in the 12% who experienced recurrences around the pancreas, the recurrences continued to occur in the Baltimore triangle.
To further lower recurrence rates, Narang and his colleagues decided to deliver an additional round of Baltimore triangle-targeted radiation to patients during surgery after removal of the pancreatic tumour. He explained that, during the surgery, surgeons remove a part of the duodenum, next to the pancreas, making it easier to access the Baltimore triangle without risking harm to surrounding organs. The combination of radiation targeted to the Baltimore Triangle prior to surgery as well as intraoperative radiation to the triangle during surgery allowed Narang to deliver ablative doses of radiation to this region.
“The combination of intraoperative radiation and targeting the Baltimore triangle has gotten us to a 5% recurrence rate, which is the lowest-ever reported recurrence rate around the pancreas for this population of patients to our knowledge. But I think we can drop to 0% in our next study,” Narang says. “We must do whatever we can to prevent recurrences from happening, because when pancreatic cancer comes back, it is often incurable. These results give us hope, though, that this can be done for a cancer where even decade ago, most thought this wasn’t possible.”
The only recurrence in the study occurred in the part of the Baltimore triangle that the team had difficulty reaching during the intraoperative treatment. Currently, the team is developing strategies to target this hard-to-reach part of the triangle, with the hopes of reducing recurrences to zero. Once they’ve mastered that refined approach, they would like to team up with other cancer centres across the US to run a larger clinical trial to confirm their results.
Source: Johns Hopkins Medicine