Ulcerative Colitis Treatment Fixes Inflammation in Gut Microbiota
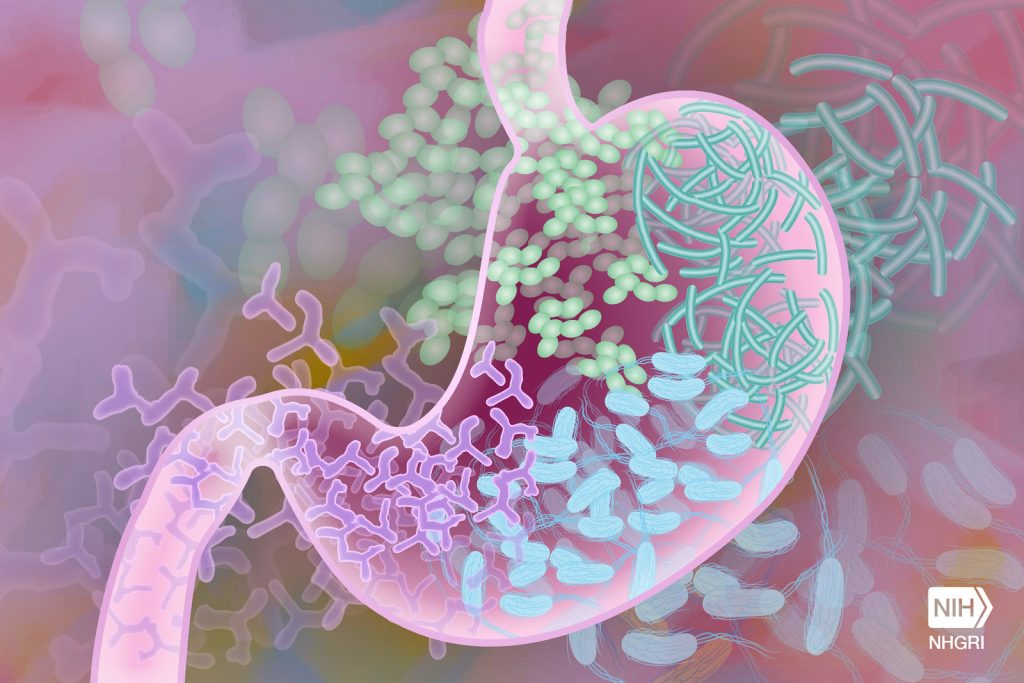
Researchers have developed a new oral treatment for ulcerative colitis that takes the innovative approach of focusing on reducing inflammation in gut microbiota.
Published in Pharmaceutics, the study comprised a two-step approach to fighting ulcerative colitis. First, the researchers reduced inflammation in gut microbiota from a mouse using an anti-inflammatory drug candidate delivered by lipid nanoparticles. Then, they orally administered the end products of these treated microbiota to the same mouse, resulting in a new, effective way to prevent ulcerative colitis.
Studies have shown that irregular gut microbiota composition is linked to ulcerative colitis, and altering this composition can effectively treat a variety of chronic diseases, including ulcerative colitis. However, current methods such as faecal microbiota transplants carry a serious infection risk because they involve the transmission of drug-resistant organisms.
In this study, the researchers developed an organism-free strategy in which gut microbiota were altered in test tubes, and then microbiota-secreted metabolites were transferred back to the host. Analysis of faeces from mice with ulcerative colitis, researchers found that a natural lipid nanoparticle-encapsulated drug candidate modified the composition of inflamed gut microbiota, which were cultured outside of the host, and the secreted metabolites.
The researchers found that their M13/nLNP nano formulation shifted the inflamed microbiota composition toward being non-inflamed. This altered microbiota composition induced significant changes in secreted metabolites, and when these metabolites were fed to mice, they established strong protection against the formation of chronic inflammation.
“Our study demonstrates that modifying microbiota outside of the host using M13/nLNP effectively reshaped the microbial secreted metabolites,” commented Dr Didier Merlin, a professor at Georgia State University. “Oral transfer of these metabolites might be an effective and safe therapeutic approach for preventing chronic ulcerative colitis.”
“Our strategy to tackle the progression of ulcerative colitis might offer an alternative and complementary approach for better managing this disease,” said Dr Chunhua Yang, a research assistant professor at the Institute for Biomedical Sciences at Georgia State. “Although this study demonstrates the anti-inflammatory effects of metabolites modified outside of the organism, further investigations are required to characterise the specific bacteria that contribute to the anti-inflammatory metabolites and to identify anti-inflammatory metabolite structures.”
Source: Georgia State University