Rising CO₂ Levels are Reflected in Human Blood. Scientists Don’t Know What it Means
Alexander Larcombe, The Kids Research Institute Australia; Curtin University and Philip Bierwirth, Australian National University
Humans evolved in an atmosphere containing roughly 200–300 parts per million (ppm) of carbon dioxide (CO₂). Today, that figure sits above 420 ppm, higher than at any point in the history of our species.
We know this extra CO₂ is contributing to climate change, but could it also be changing the chemistry of our bodies?
In our recently published research we looked at two decades of information from one of the biggest health datasets in the world to start answering this question. We found some concerning trends.
What we found
We analysed blood chemistry data from the US National Health and Nutrition Examination Survey (NHANES), which collected samples from about 7000 Americans every two years between 1999 and 2020. We looked at three markers: CO₂, calcium and phosphorus.
CO₂ is mainly carried in blood in the form of bicarbonate (HCO₃⁻).
When CO₂ enters the blood, it is converted to bicarbonate. This process largely occurs inside red blood cells, and also produces hydrogen ions.
During short-term exposure to increased CO₂, this can make blood more acidic, and result in a modest increase in bicarbonate levels in the blood (to reduce acidity).
If the exposure continues for a long time the kidneys reduce the amount of bicarbonate lost in urine and also produce more bicarbonate. This has the net effect of higher bicarbonate levels in the blood, to counteract the persistent acidity.
Levels of calcium and phosphorus in the blood may also be affected, as they too play a role in regulating acidity in the blood. These processes are completely normal.
Over the 21 years from 1999 to 2020, we found that average blood bicarbonate levels rose by about 7%. Over the same period, atmospheric CO₂ concentrations rose by a similar proportion.
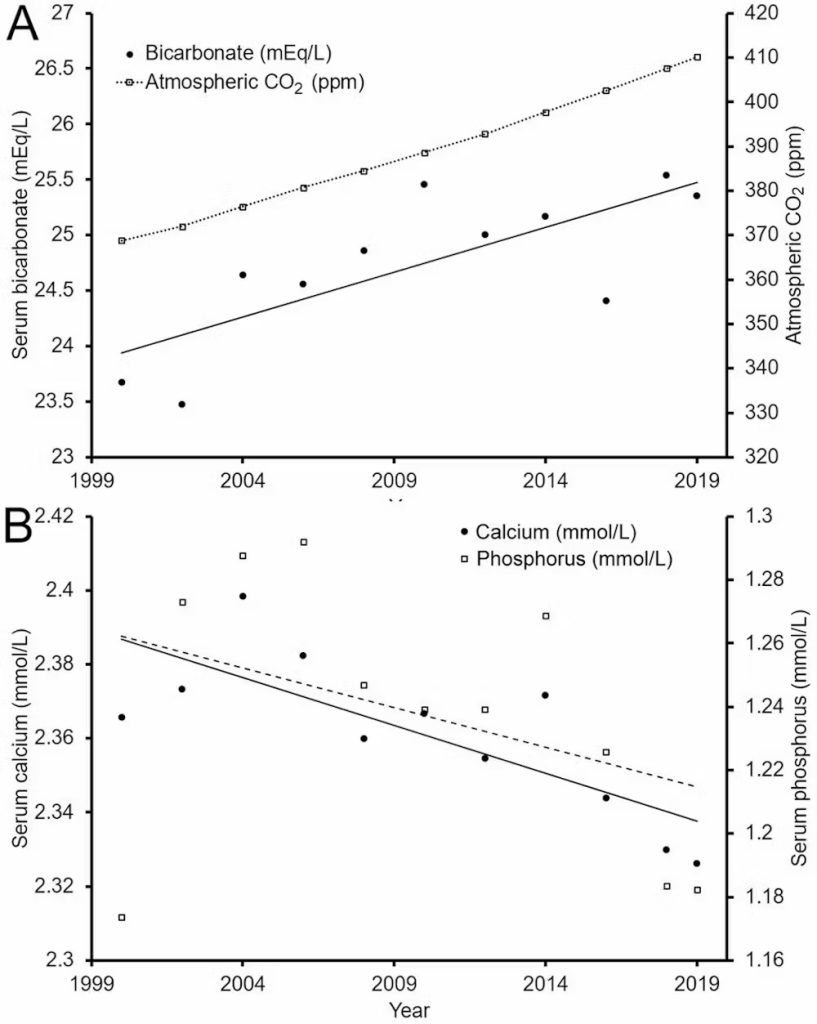
Meanwhile, blood calcium levels dropped by about 2% and phosphorus by around 7%.
If these trends continue, blood bicarbonate levels may exceed healthy levels in around 50 years. Calcium and phosphorus levels may fall below healthy levels, too, by the end of the century.
Our hypothesis is that rising CO₂ exposure could be contributing to these trends.
What’s causing the changes?
It’s important to be clear about what this study does and doesn’t show. It identifies population-level trends in blood chemistry that parallel rising atmospheric CO₂.
But correlation is not causation. The study does not adjust for factors such as diet, kidney function, diuretic use or obesity, which can influence the measurements and should be considered in future analyses.
There are other plausible contributors. One important consideration is indoor air.
Participants in the NHANES study likely spend most of their time indoors, where CO₂ concentrations often exceed 1000 ppm in poorly ventilated spaces. Other studies show time spent indoors has increased over the past two decades.
The NHANES dataset doesn’t capture this parameter, so we can’t directly assess this contribution. However, if more time indoors is contributing, it means total CO₂ exposure is rising even faster than atmospheric trends suggest. This arguably reinforces rather than alleviates the concern.
Other factors, such as shifting dietary patterns, changing rates of obesity, differences in physical activity and even variations in sample collection or processing across survey cycles, could also be important.
Can our bodies cope?
Some critics have argued that, based on what we know about how our bodies manage blood chemistry, we should have no trouble compensating for future increases in atmospheric CO₂, even under worst-case climate scenarios. For example, the lungs can increase ventilation and the kidneys can adjust to produce more bicarbonate.
For most healthy individuals, small long-term increases in outdoor CO₂ are not expected to meaningfully change the levels of bicarbonate, calcium or phosphorus in the blood.
This makes the population-level trends we observed puzzling. They could reflect a confounding rather than a direct CO₂ effect, but they do highlight how little data we have on long-term, real-world exposure.
A lack of long-term data
The argument that we can cope easily with higher CO₂ is based on short-term responses. Whether the same reasoning applies when CO₂ levels are higher across a person’s entire life remains largely untested.
There is, however, a growing body of evidence across many species which shows that even modest, environmentally relevant increases in CO₂ can produce subtle but measurable physiological effects.
In humans, short-term exposure at concentrations commonly found indoors (1000–2500 ppm) has been linked to reduced cognitive performance and changes in brain activity, though the mechanisms aren’t fully understood.
These new findings highlight a gap in evidence about long-term, real-world CO₂ exposure and human physiology. Unfortunately, there simply aren’t any studies assessing the physiological effects of breathing slightly elevated CO₂ over a lifetime.
This is particularly important for children, who will experience the longest cumulative exposure. And that’s why it’s vital to investigate this area further.
What this means
Our findings are not suggesting people will become suddenly unwell when atmospheric CO₂ reaches a certain level. What the data show is a signal that warrants attention.
If rising atmospheric CO₂ is contributing to gradual shifts in blood chemistry at a population level, then the composition of the atmosphere should be monitored alongside traditional climate indicators as a potential factor in long-term public health.
Reducing CO₂ emissions remains crucial for limiting global warming. Our findings suggest it may also be important for safeguarding aspects of human health that we’re only just beginning to understand.
Alexander Larcombe, Associate Professor and Head of Respiratory Environmental Health, The Kids Research Institute Australia; Curtin University and Philip Bierwirth, Emeritus Research Associate, Australian National University
This article is republished from The Conversation under a Creative Commons license. Read the original article.