Under Different Anaesthetics, Same Result: Unconsciousness by Shifting Brainwave Phase
MIT study finds that an easily measurable brain wave shift may be a universal marker of unconsciousness under anaesthesia
At the level of molecules and cells, ketamine and dexmedetomidine work very differently, but in the operating room, they do the same exact thing: anaesthetise the patient. By demonstrating how these distinct drugs achieve the same result, a new study in animals by neuroscientists at The Picower Institute for Learning and Memory at MIT identifies a potential signature of unconsciousness that is readily measurable to improve anaesthesiology care.
What the two drugs have in common, the researchers discovered, is the way they push around brain waves, which are produced by the collective electrical activity of neurons. When brain waves are in phase, meaning the peaks and valleys of the waves are aligned, local groups of neurons in the brain’s cortex can share information to produce conscious cognitive functions such as attention, perception and reasoning, said Picower Professor Earl K. Miller, senior author of the new study in Cell Reports. When brain waves fall out of phase, local communications, and therefore functions, fall apart, producing unconsciousness.
The finding, led by graduate student Alexandra Bardon, not only adds to scientists’ understanding of the dividing line between consciousness and unconsciousness, Miller said, but also could provide a common new measure for anesthesiologists who use a variety of different anesthetics to maintain patients on the proper side of that line during surgery.
“If you look at the way phase is shifted in our recordings, you can barely tell which drug it was,” said Miller, a faculty member in The Picower Institute and MIT’s Department of Brain and Cognitive Sciences. “That’s valuable for medical practice. Plus if unconsciousness has a universal signature, it could also reveal the mechanisms that generate consciousness.”
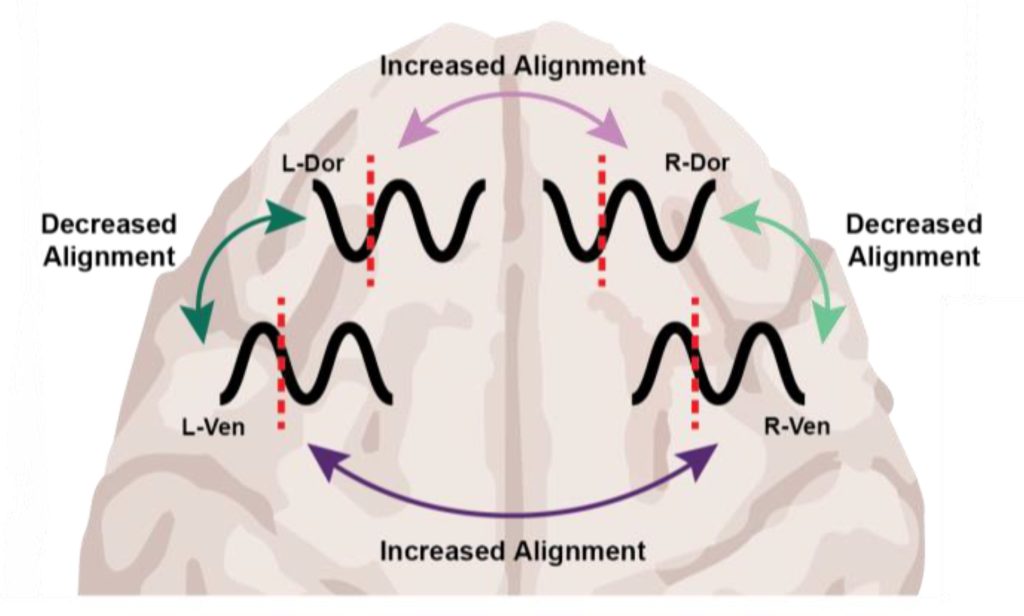
If more anesthetic drugs are also shown to affect phase in the same way, then anaesthesiologists might be able to use brain wave phase alignment as a reliable marker of unconsciousness as they titrate doses of anesthetic drugs, Miller said, regardless of which particular mix of drugs they are using. That insight could aid efforts to build closed-loop systems that can aid anaesthesiologists by constantly adjusting drug dose based on brain wave measurements of the patient’s unconsciousness.
Miller has been collaborating with study co-author Emery N. Brown, an anaesthesiologist and Professor of Computational Neuroscience and Medical Engineering, on building such a system. In a recent clinical trial with colleagues in Japan, Brown demonstrated that monitoring brain wave power signals using EEG enabled an anaesthesiologist to use much less sevoflurane during surgery with young children. The reduced doses proved safe and were associated with many improved clinical outcomes, including a reduced incidence of post-operative delirium.
Phase findings
Neuroscientists studying anaesthesia have rarely paid attention to phase, but in the new study, Bardon, Brown and Miller’s team made a point of it as they anaesthetised two animals.
After the animals lost consciousness, the measurements indicated a substantial increase in “phase locking,” especially at low frequencies. Phase locking means that the relative differences in phase remained more stable. But what caught the researchers’ attention were the differences that became locked in: Within each hemisphere, regardless of which anesthetic they used, brain wave phase became misaligned between the dorsolateral and ventrolateral regions of the prefrontal cortex.
Surprisingly, brain wave phase across hemispheres became more aligned, not less. But Miller notes that case is still a big shift from the conscious state, in which brain hemispheres are typically not aligned well, so the finding is a further indication that major changes of phase alignment, albeit in different ways at different distances, are a correlate of unconsciousness compared to wakefulness.
“The increase in interhemispheric alignment of activity by anesthetics seems to reverse the pattern observed in the awake, cognitively engaged brain,” the Bardon and Miller team wrote in Cell Reports.
Determined by distance
Distance proved to be a major factor in determining the change in phase alignment. Even across the 2.5 millimetres of a single electrode array, low-frequency waves moved 20-30 degrees out of alignment. Across the 20 or so millimetres between arrays in the upper (dorsolateral) and lower (ventrolateral) regions within a hemisphere, that would mean a roughly 180-degree shift in phase alignment, which is a complete offset of the waves.
The dependence on distance is consistent with the idea of waves traveling across the cortex, Miller said. Indeed in a 2022 study, Miller and Brown’s labs showed that the anaesthetic propofol induced a powerful low-frequency traveling wave that swept straight across the cortex, overwhelming higher-frequency straight and rotating waves.
The new results raise many opportunities for follow-up studies, Miller said. Does propofol also produce this signature of changed phase alignment? What role do travelling waves play in the phenomenon? And given that sleep is also characterised by increased power in slow wave frequencies, but is definitely not the same state as anaesthesia-induced unconsciousness, could phase alignment explain the difference?
Source: Picower Institute