Alzheimer’s Risk Gene APOE4 Silently Undermines Bone Quality in Women
Buck Institute researchers discover a surprising connection between a major risk factor for Alzheimer’s

Scientists at the Buck Institute for Research on Aging, along with collaborators at UC San Francisco, have discovered that APOE4, the most common genetic risk factor for Alzheimer’s disease, causes bone quality deficits specifically in female mice, through a mechanism that is invisible to standard imaging and can emerge as early as midlife.
The findings, published in Advanced Science, reveal an unexpected biological link between Alzheimer’s risk and skeletal health, and identify a new molecular pathway that could one day inform earlier diagnosis of cognitive decline or guide treatment for bone quality loss in women who carry the APOE4 gene.
“What makes this finding so striking is that bone quality is being compromised at a molecular level that a standard bone scan simply will not catch,” says Buck professor Birgit Schilling, PhD, a senior author of the study. “APOE4 is quietly disrupting the very cells responsible for keeping bone strong, and it is doing this specifically in females, which mirrors what we see with Alzheimer’s disease risk.”
Physicians have long observed that people with Alzheimer’s disease suffer bone fractures at higher rates, and that a diagnosis of osteoporosis in women is actually the earliest known predictor for Alzheimer’s. But the underlying mechanism connecting brain and bone health has remained elusive.
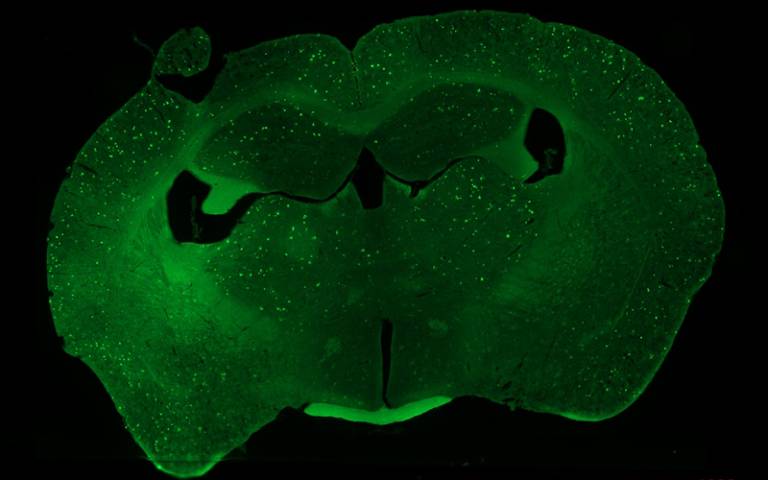
To investigate this connection, researchers, led by research scientist and co-first author of the paper Charles Schurman, PhD, first performed a proteomic analysis of aged mouse bone, a comprehensive survey of all the proteins present in the tissue. “The team discovered that bone, and particularly osteocytes, the long-lived cells embedded within it, is unusually rich in proteins associated with neurological disease, including apolipoprotein E [APOE] and amyloid precursor protein,” says Schurman. “Notably, APOE expression in osteocytes was twice as high in aged female mice as in young or male mice.”
The team then turned to a humanised mouse model carrying either APOE2 (associated with reduced Alzheimer’s risk), APOE3 (considered neutral), or APOE4 (the risk variant), and analysed bone and hippocampal tissue from the same animals. APOE4 produced strong, sex-specific effects on both the bone transcriptome and proteome; researchers found the protein-level disruption in bone was actually more pronounced than the corresponding changes in the hippocampus.
Despite the protein level disruption, cortical bone structure appeared normal under imaging. Researchers found that bone quality deficits arose not from changes in bone shape or density, but from APOE4’s suppression of perilacunar/canalicular remodelling, the process by which osteocytes actively maintain the microscopic channels that keep bone mechanically resilient. When this maintenance breaks down, bone quality deteriorates even when it looks intact.
“These results suggest that osteocytes could serve as early biological sentinels for age-related cognitive decline in women carrying APOE4,” says professor Lisa Ellerby, PhD, also a senior author of the paper. The Ellerby lab studies genetic risk factors for Alzheimer’s. “We think that targeting osteocyte function may open a new front in preserving bone quality in this population.”
Researchers say there is a larger takeaway from this research that links brain and bone science. “While we think this work is relevant for human patients with Alzheimer’s disease or with osteoporosis, this study also highlights the need for researchers to consider the human body as an entire system without isolating organs and diseases from each other,” says Ellerby.