Early Treatment Can Delay Rheumatoid Arthritis for Years
Early treatment for people at high risk of developing rheumatoid arthritis can delay the disease for several years.

Treating people who are at high risk of rheumatoid arthritis (RA) can delay the onset of the disease for several years, with benefits also continuing well after treatment has stopped. A new trial has shown that one year of treatment with the drug abatacept, a biologic therapy that targets immune cell activation, reduced progression to rheumatoid arthritis in people at high risk.
The new King’s College London study, published in The Lancet Rheumatology, builds on results from a trial reported by King’s researchers in 2024.
While the original trial followed 213 participants from the UK and the Netherlands for two years, the new study reports outcomes from an extended follow-up period of between four and eight years, making it one of the longest follow-up studies of its kind in people at risk of RA.
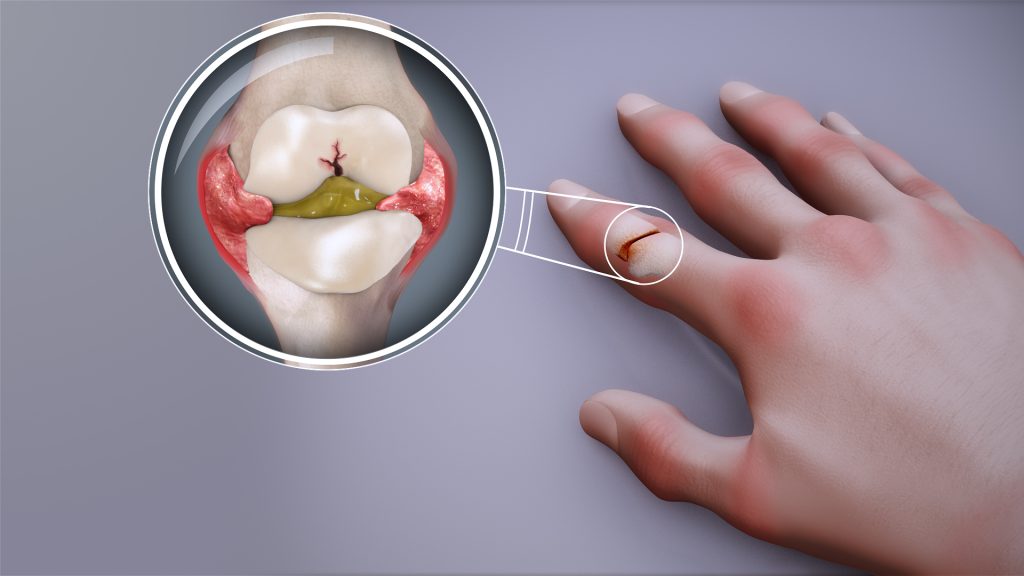
RA is a chronic autoimmune condition affecting around half a million people in the UK. It develops when the immune system mistakenly attacks the joints, causing pain, swelling, fatigue and long-term disability.
People at risk of developing the condition also often stop working before the disease starts, which creates employment instability and has economic consequences.
While effective treatments exist for people with established RA, there is currently no licensed therapy that can prevent the disease from developing in those at risk.
The researchers found that the benefits of just 12 months of abatacept therapy persisted well beyond the treatment period. People who received the drug took significantly longer to develop RA than those given placebo, with disease onset delayed by up to four years beyond the treatment period.
Although the drug did not permanently prevent RA, the findings show that early treatment can alter the course of the disease by postponing its development, potentially reducing the number of years people live with symptoms and complications.
Intervening early in people at high risk of RA can have lasting benefits. We have shown that this approach is safe and can prevent disease while patients are on treatment as well as substantially relieve symptoms. Importantly, it can also delay the onset of RA for several years, even after treatment has stopped. This could reduce how long people live with symptoms and complications, drastically improving their quality of life.
Professor Andrew Cope, Professor of Rheumatology in the Centre for Rheumatic Diseases at King’s College London
The study also showed that abatacept was most effective in individuals at highest risk of developing RA, identified through a blood test detecting specific autoantibodies. While these participants were at highest risk of progressing to RA they were also much more likely to benefit from early intervention.
During the at-risk phase, treatment with abatacept reduced symptoms such as joint pain and fatigue and improved overall wellbeing. But once treatment stopped, symptom levels became similar between the treatment and placebo groups, suggesting that continued immune modulation may be required to sustain symptom control.
The study found that abatacept was safe, with similar rates of serious adverse events in both the treatment and placebo groups, and no safety concerns linked to the drug.
The researchers suggest the latest findings provide evidence that early, targeted immune treatment can delay RA in people at highest risk, supporting further research into preventive approaches for autoimmune disease.
Source: King’s College London