Scientists Regenerate Joint Cartilage in Mice by Blocking an Ageing Protein
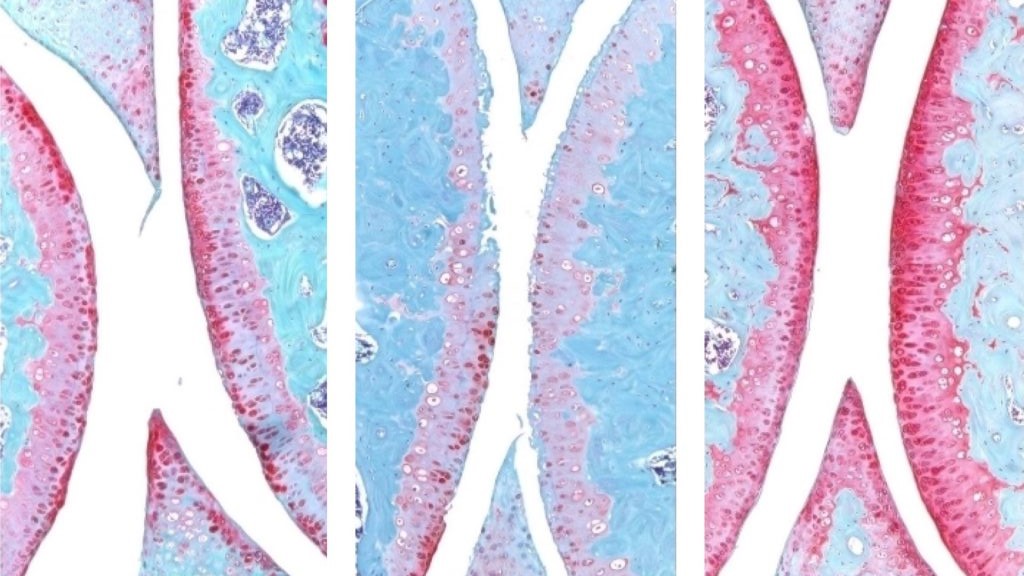
An injection that blocks the activity of a protein involved in ageing reverses naturally occurring cartilage loss in the knee joints of old mice, a Stanford Medicine-led study has found. The treatment also prevented the development of arthritis after knee injuries mirroring the ACL tears often experienced by athletes or recreational exercisers. An oral version of the treatment is already in clinical trials with the goal of treating age-related muscle weakness.
Samples of human tissue from knee replacement surgeries – which include both the extracellular scaffolding, or matrix, in the joint as well as cartilage-generating chondrocyte cells – also responded to the treatment by making new, functional cartilage.
The study results suggest it may be possible to regenerate cartilage lost to ageing or arthritis with an oral drug or local injection, rendering knee and hip replacement unnecessary.
The treatment directly targets the cause of osteoarthritis, a disease for which no drug can slow down or reverse its progress; the primary treatments for osteoarthritis are pain control and surgical replacement of the affected joints.
The protein, 15-PGDH – termed a gerozyme due to its increase in prevalence as the body ages – is a master regulator of ageing. Gerozymes, identified by the same researchers in 2023, also drive the loss of tissue function. They are a major force behind age-related loss of muscle strength in mice. Blocking the function of 15-PGDH with a small molecule results in an increase in old animals’ muscle mass and endurance. Conversely, expressing15-PGDH in young mice causes their muscles to shrink and weaken. The gerozyme has also been implicated in the regeneration of bone, nerve and blood cells.
In each of these tissues, regeneration is due to increases in the proliferation and specialisation of tissue-specific stem cells. However, chondrocytes change their patterns of gene expression to assume a more youthful state without the involvement of stem cells.
“This is a new way of regenerating adult tissue, and it has significant clinical promise for treating arthritis due to ageing or injury,” said Helen Blau, PhD, professor of microbiology and immunology. “We were looking for stem cells, but they are clearly not involved. It’s very exciting.”
Blau, who directs the Baxter Laboratory for Stem Cell Biology and Nidhi Bhutani, PhD, associate professor of orthopaedic surgery, are the senior authors of the research, which was published online in Science.
‘Dramatic regeneration’
“Millions of people suffer from joint pain and swelling as they age,” Bhutani said. “It is a huge unmet medical need. Until now, there has been no drug that directly treats the cause of cartilage loss. But this gerozyme inhibitor causes a dramatic regeneration of cartilage beyond that reported in response to any other drug or intervention.”
There are three main types of cartilage in the human body. One, elastic cartilage, is soft and flexible and forms structures like the outer ear. A second, fibrocartilage, is dense and tough, absorbing shock in areas such as between the spinal vertebrae. The third, hyaline cartilage, is smooth and glossy, providing a low-friction surface for lubrication and flexibility in joints like the ankles, hips, shoulders and parts of the knee. Hyaline cartilage, also known as articular cartilage, is the cartilage most commonly affected by osteoarthritis.
Osteoarthritis occurs when a joint is stressed by ageing, injury or obesity. The chondrocytes begin to release pro-inflammatory molecules and to break down collagen, which is the primary structural protein of cartilage. When collagen is lost, the cartilage thins and softens; the accompanying inflammation causes the joint swelling and pain that are hallmarks of the disease. Under normal circumstances, articular cartilage rarely regenerates. Although some populations of putative stem or progenitor cells capable of generating cartilage have been identified in bone, attempts to identify similar populations of cells in the articular cartilage have been unsuccessful.
Previous research from Blau’s lab has shown that a molecule called prostaglandin E2 is essential to muscle stem cell function. 15-PGDH degrades prostaglandin E2. Inhibiting 15-PGDH activity, or increasing levels of prostaglandin E2, supports the regeneration of damaged muscle, nerve, bone, colon, liver and blood cells in young mice.
Blau, Bhutani and their colleagues wondered if 15-PGDH might also play a role in ageing cartilage and joints. They wanted to find out if a similar pathway contributes to cartilage loss from ageing or in response to injury. When they compared the amount of 15-PGDH in the knee cartilage in young versus old mice, they saw that, as in other tissues, levels of the gerozyme increased about two-fold with age.
They next experimented with injecting old animals with a small molecule drug that inhibits 15-PGDH activity – first into the abdomen, which affects the entire body, then directly into the joint. In each case, the knee cartilage, which was markedly thinner and less functional in older animals as compared with younger mice, thickened across the joint surface. Further experiments confirmed that the chondrocytes in the joint were generating hyaline, or articular, cartilage, rather than less-functional fibrocartilage.
“Cartilage regeneration to such an extent in aged mice took us by surprise,” Bhutani said. “The effect was remarkable.”
Addressing ACL tears
Similar results were observed in animals with knee injuries like the ACL tears that frequently occur in people participating in sports such as soccer, basketball and skiing that require sudden pivoting, stopping or jumping. While the tears can be surgically repaired, about 50% of people develop osteoarthritis in the injured joint within about 15 years.
The researchers found that a series of injections twice a week for four weeks of the gerozyme inhibitor after injury dramatically reduced the chance that osteoarthritis develops in the mice. Animals treated with a control drug had levels of 15-PGDH that were twice as high as in their uninjured peers, and they developed osteoarthritis within four weeks.
The animals treated with the gerozyme inhibitor also moved more typically and put more weight on the paw of the affected leg than did untreated animals.
“Interestingly, prostaglandin E2 has been implicated in inflammation and pain,” Blau said. “But this research shows that, at normal biological levels, small increases in prostaglandin E2 can promote regeneration.”
A closer investigation of the chondrocytes in the joints of old mice and young mice showed that old chondrocytes expressed more detrimental genes involved in inflammation and the conversion of hyaline cartilage to unwanted bone, and fewer genes involved in cartilage development.
The researchers were also able to pinpoint subcategories of old chondrocytes that change their patterns of gene expression after treatment. One, which expresses 15-PGDH and genes involved in cartilage degradation, decreased in prevalence from 8% to 3% after treatment. Another, which does not express 15-PGDH but does express genes involved in the production of fibrocartilage, also decreased in prevalence: from 16% to 8% after treatment. A third population, which does not make 15-PGDH and which expresses genes involved in hyaline cartilage formation and the maintenance of the extracellular matrix necessary for its function, increased in prevalence after treatment from 22% to 42%. The findings indicate an overall shift in gene expression after treatment to a more youthful cartilage composition – without the involvement of stem or progenitor cells.
Finally, the researchers studied human cartilage tissue removed from patients with osteoarthritis undergoing total knee replacements. Tissue treated with the 15-PGDH inhibitor for one week exhibited lower levels of 15-PGDH-expressing chondrocytes and lowered cartilage degradation and fibrocartilage genes than control tissue and began to regenerate articular cartilage.
“The mechanism is quite striking and really shifted our perspective about how tissue regeneration can occur,” Bhutani said. “It’s clear that a large pool of already existing cells in cartilage are changing their gene expression patterns. And by targeting these cells for regeneration, we may have an opportunity to have a bigger overall impact clinically.”
Blau added, “Phase 1 clinical trials of a 15-PGDH inhibitor for muscle weakness have shown that it is safe and active in healthy volunteers. Our hope is that a similar trial will be launched soon to test its effect in cartilage regeneration. We are very excited about this potential breakthrough. Imagine regrowing existing cartilage and avoiding joint replacement.”
Source: Stanford University



