How One ‘Forever Chemical’ Can Disrupt Foetal Facial Development
Researchers discovered perfluorodecanoic acid is the most toxic during foetal craniofacial development
Researchers have long associated per- and polyfluoroalkyl substances (PFAS), commonly known as “forever chemicals,” to certain severe birth defects but exactly how these pollutants harm a developing foetus has remained mostly a mystery. New research now provides the first clear molecular explanation, showing how one PFAS, called perfluorodecanoic acid (PFDA), can trigger craniofacial abnormalities before birth.
The research was published in ACS Chemical Research in Toxicology.
“Most people are exposed to small amounts of PFAS in everyday life but higher exposure can occur through contaminated water, living near manufacturing sites or certain jobs like firefighting and ski waxing, which is why it’s so important to understand the chemicals better,” said the paper’s senior author Jed Lampe, PhD, associate professor at University of Colorado Anschutz Skaggs School of Pharmacy and Pharmaceutical Sciences. “We wanted to understand which PFAS compounds are truly harmful during foetus development, especially for people with higher exposure, and how they cause damage.”
There are approximately 15 000 PFAS used in consumer and industrial products but scientists are increasingly finding that only some pose serious health risks. In this study, Lampe worked with the paper’s first author Michaela Hvizdak and co-author Sylvie Kandel to test 13 commonly found PFAS and discovered PFDA as the most toxic during foetal craniofacial development.
They demonstrated a possible link between PFDA and the extensive facial changes observed in humans and laboratory animals, with some estimates indicating a 10% increased risk at extremely low exposure levels.
“This finding moves us beyond association by providing a clear explanation for how PFDA can interfere with foetal development. It’s a critical step toward understanding a vast and complex class of environmental chemicals,” said Lampe.
The cause of underdeveloped eyes and an abnormal jaw
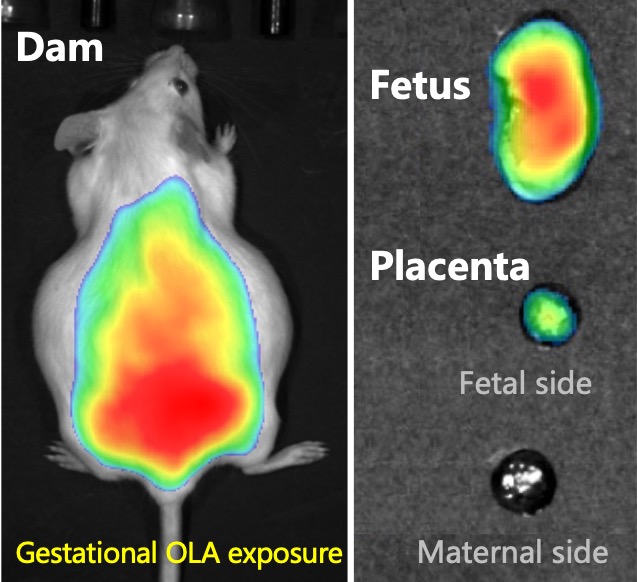
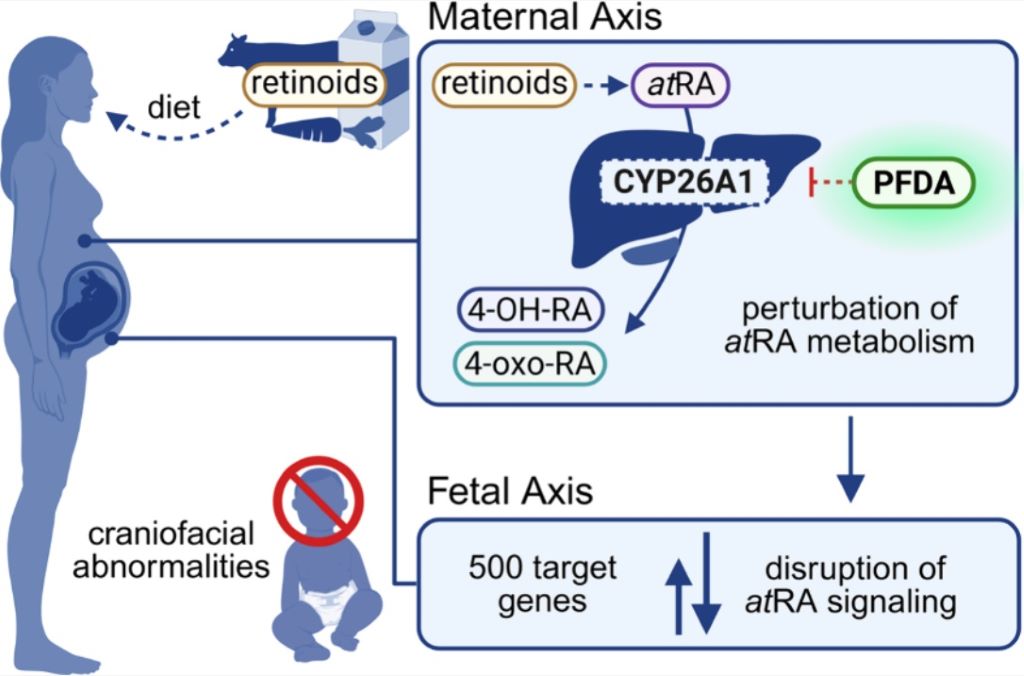
They found that PFAS disrupts retinoic acid, a molecule essential for shaping the face and head during early pregnancy. Retinoic acid regulates hundreds of genes and its levels must be controlled. Because a foetus cannot produce or safely eliminate excess retinoic acid, it relies entirely on the mother to maintain the homeostatic balance of the hormone.
The researchers discovered PFDA blocks CYP26A1, a key enzyme responsible for breaking down excess retinoic acid. When this enzyme is inhibited, retinoic acid levels can rise too high, disrupting normal facial development. PFDA also suppresses the genes that produce this enzyme through a separate biological pathway, delivering a “double hit” to the system that regulates early development.
“As a result, severe craniofacial abnormalities can develop, including underdeveloped eyes and abnormal jaw formation, which were the most common effects of PFDA exposure during foetal development,” said Lampe.
The researchers hope by providing a molecular explanation for this abnormality that the research and scientific community can work to develop targeted laboratory assays and computer‑based screening tools to quickly rank PFAS by risk. They say this could help regulators and manufacturers distinguish more dangerous compounds from safer alternatives and guide the design of less toxic chemicals in the future.
They also hope the research can open the door to practical interventions for people with higher exposure, such as firefighters and ski wax technicians. This could include looking into how to reduce exposure levels and whether strategies can be developed to lower PFDA during pregnancy to protect foetal development.
By Julia Milzer
Source: CU Anschutz Medical Campus