Hydraulic Brain: Body Motion Linked to Fluid Movement in the Brain
Abdominal contractions are tightly linked to gentle brain movements that help circulate CSF
The brain is more mechanically connected to the body than previously appreciated, scientists reported in Nature Neuroscience. Through a study using mice and simulations, the team found a potential biological mechanism underlying why exercise is thought to benefit brain health: abdominal contractions compress blood vessels connected to the spinal cord and the brain, enabling the organ to gently move within the skull. This swaying facilitates the surrounding cerebrospinal fluid to flow over the brain, potentially washing away neural waste that could cause problems for brain function.
According to Patrick Drew, professor of engineering science and mechanics, of neurosurgery, of biology and of biomedical engineering at Penn State, the work builds on previous studies detailing how sleep and neuron loss can influence how and when cerebrospinal fluid flushes through the brain.
“Our research explains how just moving around might serve as an important physiological mechanism promoting brain health,” said Drew, corresponding author on the paper. “In this study, we found that when the abdominal muscles contract, they push blood from the abdomen into the spinal cord, just like in a hydraulic system, applying pressure to the brain and making it move. Simulations show that this gentle brain movement will drive fluid flow in and around the brain. It is thought the movement of fluid in the brain is important for removing waste and preventing neurodegenerative disorders. Our research shows that a little bit of motion is good, and it could be another reason why exercise is good for our brain health.”
Drew, who also holds the title of associate director of the Huck Institutes of the Life Sciences, explained how in a hydraulic system, a pump creates pressure that drives fluid flow. In this case, the pump is the abdominal contraction – which can be as light as the tensing prior to sitting up or taking a step. The contraction puts pressure on the vertebral venous plexus, a network of veins that connect the abdominal cavity to the spinal cavity, causing the brain to move.
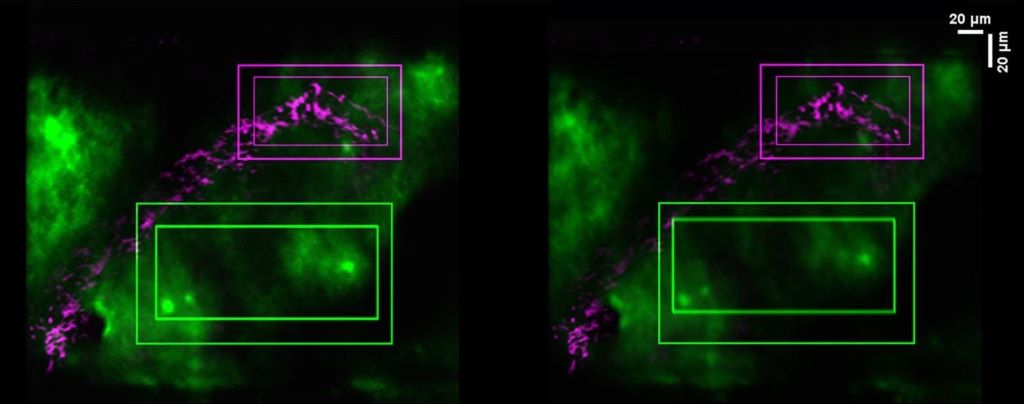
The researchers visualised the process in moving mice with two advanced imaging technologies: two-photon microscopy, which allows for high-definition imaging of living tissue, and microcomputed tomography, which enables high-resolution 3D examination of whole organs. They observed the brain shifting in the moments before the mouse moved, but right after the tightening of the abdominal muscles needed to spur the body into further movement.
To confirm that it was abdominal contractions rather than other movement that acted as the pump, the researchers applied gentle and controlled pressure to the abdomens of lightly anaesthetised mice. With no other movement other than a localised mechanical pressure less than a human would experience with a blood pressure cuff, the mice’s brains shifted.
“Importantly, the brain began moving back to its baseline position immediately upon relief of the abdominal pressure,” Drew said. “This suggests that abdominal pressure can rapidly and significantly alter the position of the brain within the skull.”
With the abdominal contraction-brain movement link confirmed, Drew said the next step was to understand the fluid’s movement in the brain and if the brain’s movement could induce fluid flow. However, there previously were no existing imaging techniques to visualize the rapid, nuanced dynamics of such fluid flows.
“Luckily, our interdisciplinary team at Penn State was able to develop these techniques, including conducting the imaging experiments of living mice and creating computer simulations of fluid motion,” Drew said. “That combination of expertise is so important for understanding these types of complicated systems and how they impact health.”
Francesco Costanzo, professor of engineering science and mechanics, of biomedical engineering, of mechanical engineering and of mathematics, led the computational modelling.
“Modelling fluid flow in and around the brain offers unique challenges because there are simultaneous, independent movements, as well as time-dependent, coupled movements. Accounting for all of them requires accounting for the special physics that happens every time a fluid particle crosses one of the many membranes in the brain,” Costanzo said. “So, we simplified it. The brain has a structure similar to a sponge, in the sense that you have a soft skeleton and fluid can move through it.”
By simplifying the geometry of the brain to that of a sponge, Costanzo explained that the team could model how fluid flows through a structure with varied spaces, like wrinkles in the brain, or pores in the sponge.
“Keeping with the idea of the brain as a sponge, we also thought of it as a dirty sponge – how do you clean a dirty sponge?” Costanzo asked. “You run it under a tap and squeeze it out. In our simulations, we were able to get a sense of how the brain moving from an abdominal contraction can help induce fluid flow over the brain to help clear waste products.”
Drew emphasised that while more work is needed to understand the full implications in humans, this study suggests that body movement may help to cycle cerebrospinal fluid around and in the brain, removing waste and helping to protect against neurodegenerative disorders associated with waste buildup.
“This kind of motion is so small. It’s what’s generated when you walk or just contract your abdominal muscles, which you do when you engage in any physical behaviour. It could make such a difference for your brain health,” Drew said.
By Ashley WennersHerron
Source: Pennsylvania State University