Inflammation Leaves a Long-lasting Impression on Intestinal Stem Cells

Researchers at Baylor College of Medicine, the University of Michigan and collaborating institutions have discovered that inflammation in the gut leaves long-term marks on intestinal stem cells (ISCs) that reduce their ability to heal the intestine, even after inflammation has receded. This is important because it affects ISCs’ response to future challenges. The study appeared in Cell Stem Cell.
“We study graft-vs-host disease (GVHD), a major cause of mortality after bone marrow transplantation, a potentially curative therapy for many blood diseases. One of our goals is to better understand GVHD and identify strategies to control it,” said corresponding author Dr Pavan Reddy, professor and director of the Dan L Duncan Comprehensive Cancer Center at Baylor and previously at the University of Michigan.
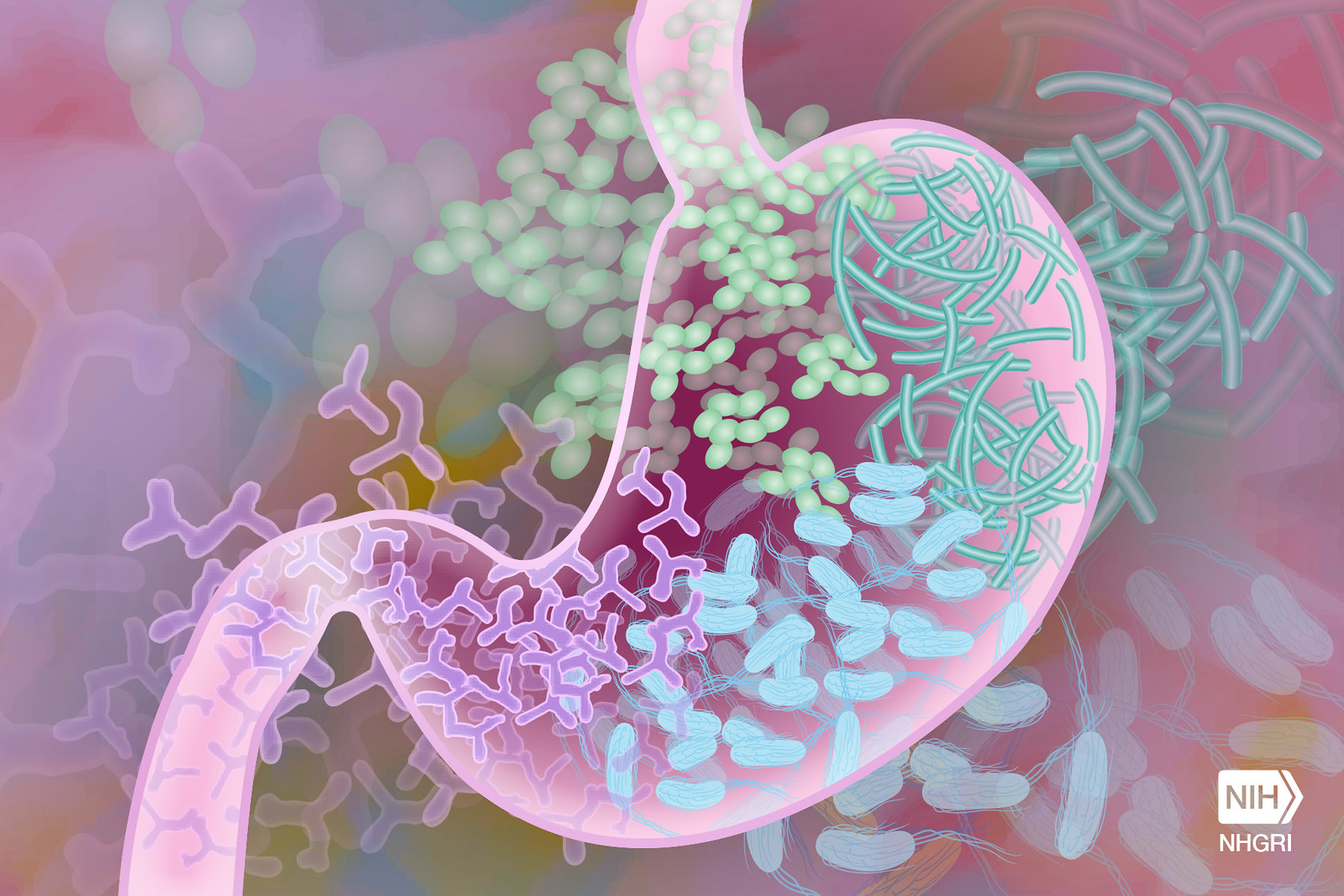
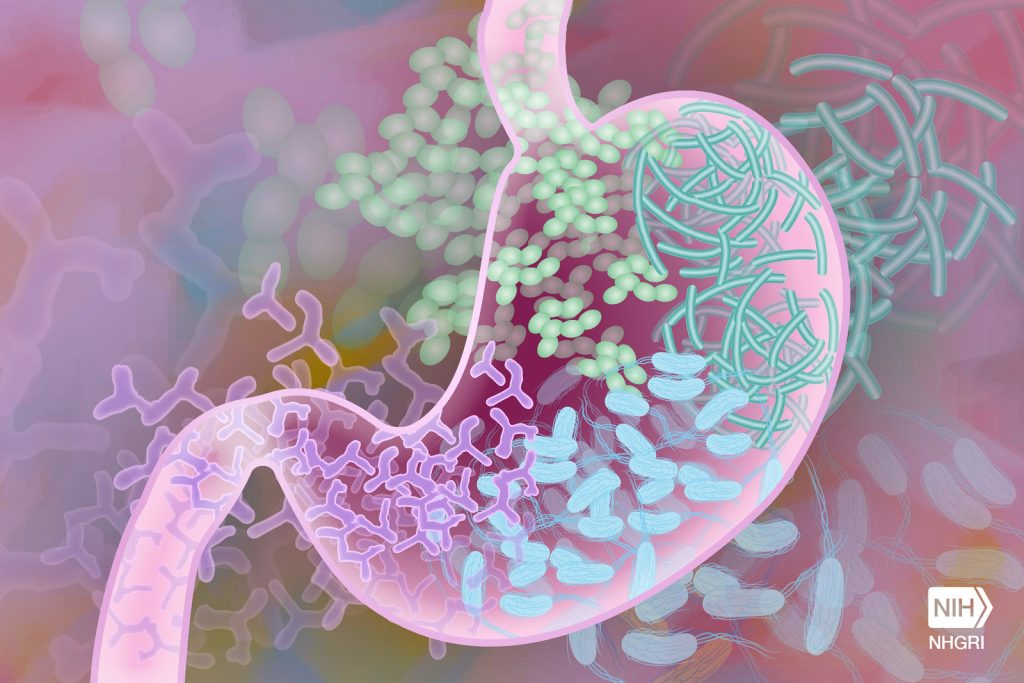
“GVHD is an inflammatory reaction in which immune T cells from the bone marrow transplant donor attack the host gut cells, mainly ISCs,” said first author Dr Dongchang Zhao, in Reddy’s lab.
Although many ISCs perish during GVHD, survivors remain. However, it’s not known whether they are fully functional or can return to their full functionality after the resolution of GVHD, which has fundamental implications for host resilience and repair.
“In the current study, we investigated the consequences of inflammation on ISCs in well-defined clinically relevant models of GVHD,” Reddy said.
“Using cellular and animal models, we found that exposure to inflammation drove ISCs to change their metabolism in ways that resulted in the accumulation of succinate, a product of cellular processes, which in turn reprogramed the epigenome,” Zhao said.
The epigenome is a system of chemical marks on the DNA that regulates the genes expressed by the cell. Inflammation-led epigenome reprogramming changed the expression of genes involved in cell reproduction. Overall, reprogrammed ISCs were less able of regenerating, a first step toward healing the intestine.
“We then investigated whether ISCs would be able to recuperate their regenerative ability after inflammation had resolved,” Reddy said. “We found that ISCs had not overcome their initial exposure to inflammation. Despite mitigating GVHD inflammation for 28 days, ISCs retained a reduced regenerative capacity that led to poor recovery and increased mortality from challenges, such as non-lethal radiation exposure, in animal models. More research is on the way to design strategies to help ISCs ‘forget’ their encounter with inflammation and enhance their resilience against immune attacks.”
Source: Baylor College of Medicine