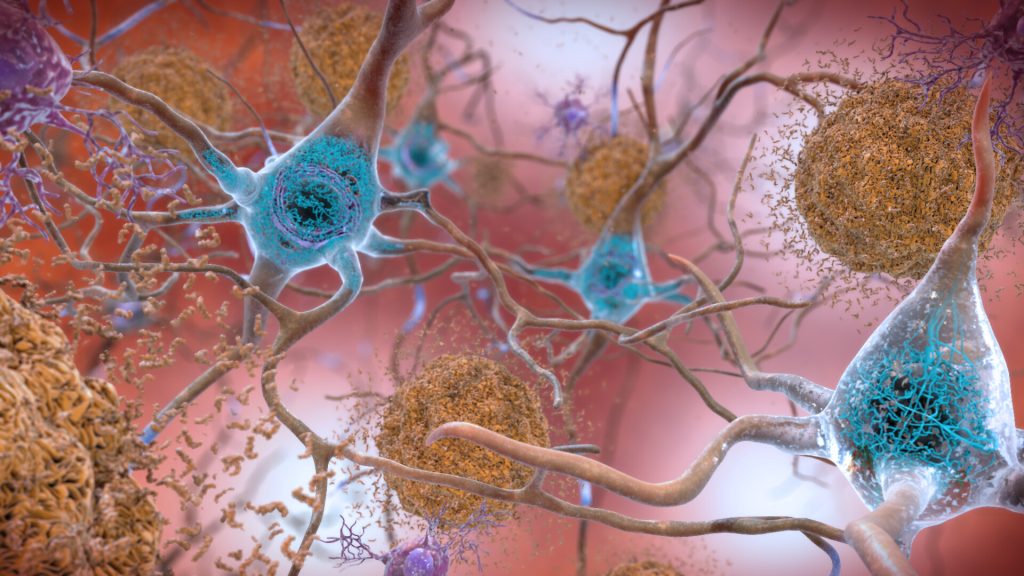
How Gamma Rhythm Light and Sound Strips Amyloid in Alzheimer’s Mouse Models
Studies at MIT and elsewhere are producing mounting evidence that light flickering and sound clicking at the gamma brain rhythm frequency of 40Hz can reduce Alzheimer’s disease (AD) progression and treat symptoms in human volunteers as well as lab mice. In a new study in Nature using a mouse model of the disease, researchers at The Picower Institute for Learning and Memory of MIT reveal a key mechanism that may contribute to these beneficial effects: clearance of amyloid proteins, a hallmark of AD pathology, via the brain’s glymphatic system, a recently discovered “plumbing” network parallel to the brain’s blood vessels.
“Ever since we published our first results in 2016, people have asked me how does it work? Why 40Hz? Why not some other frequency?” said study senior author Li-Huei Tsai, Professor of Neuroscience at Picower. “These are indeed very important questions we have worked very hard in the lab to address.”
The new paper describes a series of experiments, led by Mitch Murdock when he was a Brain and Cognitive Sciences doctoral student at MIT, showing that when sensory gamma stimulation increases 40 Hz power and synchrony in the brains of mice, that prompts a particular type of neuron to release peptides. The study results further suggest that those short protein signals then drive specific processes that promote increased amyloid clearance via the glymphatic system.
“We do not yet have a linear map of the exact sequence of events that occurs,” said Murdock, who was jointly supervised by Tsai and co-author and collaborator Ed Boyden, Professor of Neurotechnology at MIT. “But the findings in our experiments support this clearance pathway through the major glymphatic routes.”
From gamma to glymphatics
Because prior research has shown that the glymphatic system is a key conduit for brain waste clearance and may be regulated by brain rhythms, Tsai and Murdock’s team hypothesised that it might help explain the lab’s prior observations that gamma sensory stimulation reduces amyloid levels in Alzheimer’s model mice.
Working with “5XFAD” mice, which genetically model Alzheimer’s, Murdock and co-authors first replicated the lab’s prior results that 40Hz sensory stimulation increases 40Hz neuronal activity in the brain and reduces amyloid levels. Then they set out to measure whether there was any correlated change in the fluids that flow through the glymphatic system to carry away wastes. Indeed, they measured increases in cerebrospinal fluid in the brain tissue of mice treated with sensory gamma stimulation compared to untreated controls. They also measured an increase in the rate of interstitial fluid leaving the brain. Moreover, in the gamma-treated mice he measured increased diameter of the lymphatic vessels that drain away the fluids and measured increased accumulation of amyloid in cervical lymph nodes, which is the drainage site for that flow.
To investigate how this increased fluid flow might be happening, the team focused on the aquaporin 4 (AQP4) water channel of astrocyte cells, which enables the cells to facilitate glymphatic fluid exchange. When they blocked APQ4 function with a chemical, that prevented sensory gamma stimulation from reducing amyloid levels and prevented it from improving mouse learning and memory. And when, as an added test they used a genetic technique for disrupting AQP4, that also interfered with gamma-driven amyloid clearance.
In addition to the fluid exchange promoted by APQ4 activity in astrocytes, another mechanism by which gamma waves promote glymphatic flow is by increasing the pulsation of neighbouring blood vessels. Several measurements showed stronger arterial pulsatility in mice subjected to sensory gamma stimulation compared to untreated controls.
One of the best new techniques for tracking how a condition, such as sensory gamma stimulation, affects different cell types is to sequence their RNA to track changes in how they express their genes. Using this method, Tsai and Murdock’s team saw that gamma sensory stimulation indeed promoted changes consistent with increased astrocyte AQP4 activity.
Prompted by peptides
The RNA sequencing data also revealed that upon gamma sensory stimulation a subset of neurons, called “interneurons,” experienced a notable uptick in the production of several peptides. This was not surprising in the sense that peptide release is known to be dependent on brain rhythm frequencies, but it was still notable because one peptide in particular, VIP, is associated with Alzheimer’s-fighting benefits and helps to regulate vascular cells, blood flow and glymphatic clearance.
Seizing on this intriguing result, the team ran tests that revealed increased VIP in the brains of gamma-treated mice. The researchers also used a sensor of peptide release and observed that sensory gamma stimulation resulted in an increase in peptide release from VIP-expressing interneurons.
But did this gamma-stimulated peptide release mediate the glymphatic clearance of amyloid? To find out, the team ran another experiment: they chemically shut down the VIP neurons. When they did so, and then exposed mice to sensory gamma stimulation, they found that there was no longer an increase in arterial pulsatility and there was no more gamma-stimulated amyloid clearance.
“We think that many neuropeptides are involved,” Murdock said. Tsai added that a major new direction for the lab’s research will be determining what other peptides or other molecular factors may be driven by sensory gamma stimulation.
Tsai and Murdock added that while this paper focuses on what is likely an important mechanism – glymphatic clearance of amyloid – by which sensory gamma stimulation helps the brain, it’s probably not the only underlying mechanism that matters. The clearance effects shown in this study occurred rather rapidly but in lab experiments and clinical studies weeks or months of chronic sensory gamma stimulation have been needed to have sustained effects on cognition.
With each new study, however, scientists learn more about how sensory stimulation of brain rhythms may help treat neurological disorders.
Source: Picower Institute at MIT