Human Papillomavirus Infection Kinetics Revealed in New Longitudinal Study
Improved understanding of why some infections persist could improve treatment, screening, and vaccination strategies for HPV infection
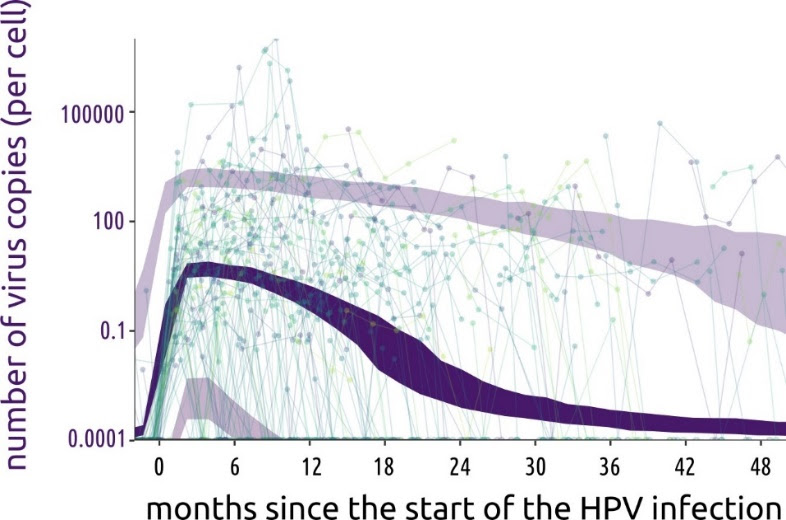
Non-persistent human papillomavirus (HPV) infections are characterized by a sharp increase in viral load followed by a long plateau, according to a study published January 21st in the open-access journal PLOS Biology by Samuel Alizon of the National Centre for Scientific Research (CNRS), France, and colleagues.
Chronic HPV infection is responsible for more than 600 000 new cancers each year, including nearly all cervical cancers. Infection among young women is common, impacting nearly 20% of women 25 years of age. Fortunately, the vast majority of these infections clear within two years without symptoms. Still, a better understanding of why some infections clear while others persist could improve treatment, screening, and vaccination strategies for HPV-associated diseases.
To monitor the dynamics of HPV infection, the PAPCLEAR cohort study in France followed 189 women aged 18 to 25 years for up to 24 months. Researchers followed participants every two months, collecting information on viral kinetics and immune markers.
By frequently monitoring participants, the researchers achieved unprecedented temporal resolution on viral and immune kinetics. They found that non-persistent infections are characterised by a plateau in HPV viral load that starts around 2 months after infection and lasts for 13 to 20 months before rapidly declining. Additionally, they found a strong correlation between a population of immune cells in between innate and adaptive immunity, namely TCRγδ cells, and the total amount of viruses produced.
The study is limited by the fact that many of the infection follow-ups were truncated, meaning that the participants were already infected when enrolled or were lost to follow-up before infection clearance. Furthermore, only 4 out of 76 infected participants were followed for 18 months or more. Therefore, differences between chronic and acute infections will have to wait for longer studies.
The authors add, “Asymptomatic genital infections by human papillomaviruses (HPVs) display striking dynamics at the frontier between acute and chronic infections. HPV infections appear to be associated with specific patterns of the innate and adaptative local immune response.”
Provided by PLOS