Scientists Find Hidden Diversity Among T. Gondii
UC Riverside study reshapes understanding of toxoplasmosis and identifies new paths for treatment
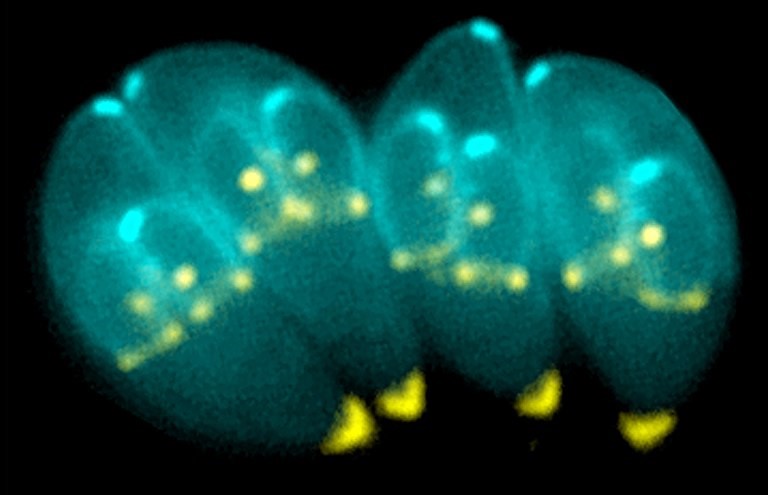
A University of California, Riverside team of scientists has found that Toxoplasma gondii, a common parasite affecting up to one-third of the global population, is far more complex than previously believed. The findings, published in Nature Communications, offer new insight into how T. gondii causes disease and why it has been so difficult to treat.
Humans commonly contract toxoplasmosis by eating undercooked meat or through exposure to contaminated soil or cat faeces. The parasite is best known for its ability to hide in the body by forming tiny cysts in the brain and muscles.
Most people who are infected never notice any symptoms. However, the parasite remains in the body for life as cysts, which can contain hundreds of parasites. The parasites they lodge can become active again later, however, especially in people with weakened immune systems, leading sometimes to serious problems affecting the brain or eyes. Most people who are infected never notice any symptoms. Infection during pregnancy can cause serious complications for developing babies with limited immune systems.
Until now, scientists believed that the cysts contained a single, uniform type of parasite lying dormant until it reactivated. But using advanced single-cell analysis techniques, the UC Riverside team discovered that each cyst contains multiple distinct subtypes of parasites, each with different biological roles.
“We found the cyst is not just a quiet hiding place – it’s an active hub with different parasite types geared toward survival, spread, or reactivation,” said Emma Wilson, a professor of biomedical sciences in the UCR School of Medicine who led the study.
Wilson explained that cysts form slowly under immune pressure and are encased in a protective wall, housing hundreds of slow-replicating parasites called bradyzoites. Although microscopic, cysts are relatively large for intracellular pathogens, reaching up to 80 microns in diameter, with each bradyzoite measuring roughly five microns in length. They reside primarily within neurons but are also commonly found in skeletal and cardiac muscle, which is important since humans are often infected by consuming undercooked meat containing these cysts.
According to Wilson, cysts are clinically and biologically significant for several reasons. They are resistant to all existing therapies and remain in the body once established. They facilitate transmission between hosts. When reactivated, bradyzoites convert into fast-replicating tachyzoites that disseminate throughout tissues, causing severe disease such as toxoplasmic encephalitis (neurological damage) or retinal toxoplasmosis (vision loss).
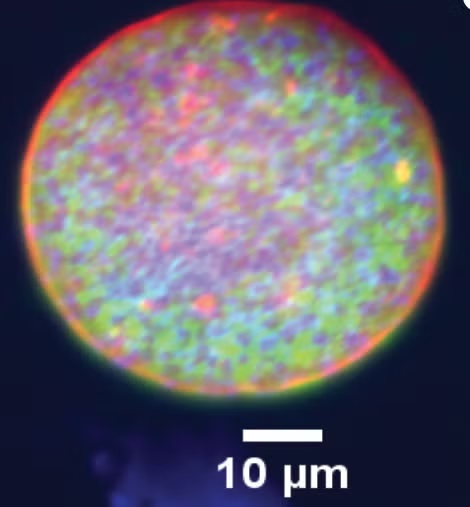
“For decades, the Toxoplasma life cycle was understood in overly simplistic terms, conceptualised as a linear transition between tachyzoite and bradyzoite stages,” Wilson said. “Our research challenges that model. By applying single-cell RNA sequencing to parasites isolated directly from cysts in vivo, we found unexpected complexity within the cyst itself. Rather than a uniform population, cysts contain at least five distinct subtypes of bradyzoites. Although all are classified as bradyzoites, they are functionally different, with specific subsets primed for reactivation and disease.”
Wilson acknowledged that studying cysts has long been a technical challenge. They grow slowly, are embedded deep within tissues like the brain, and do not form efficiently in standard laboratory cultures. As a result, most genetic and molecular studies of Toxoplasma have focused on tachyzoites grown in vitro, leaving the biology of cyst-resident bradyzoites poorly understood.
“Our work overcomes those limitations by using a mouse model that closely mirrors natural infection,” Wilson said. “Because mice are a natural intermediate host for Toxoplasma, their brains can harbour thousands of cysts. By isolating these cysts, digesting them enzymatically, and analysing individual parasites, we were able to gain a view of chronic infection as it occurs in living tissue.”
Wilson explained that current treatments for toxoplasmosis can control the fast-growing form of the parasite that causes acute illness, but no existing drugs can eliminate the cysts.
“By identifying different parasite subtypes inside cysts, our study pinpoints which ones are most likely to reactivate and cause damage,” she said. “This helps explain why past drug development efforts have struggled and suggests new, more precise targets for future therapies.”
Congenital toxoplasmosis remains a major concern when primary infection occurs during pregnancy, potentially leading to severe foetal outcomes. Although prior immunity typically protects the foetus, routine screening is lacking in some countries, reflecting how difficult it is to manage an infection that is common but usually symptom-free.
Despite its prevalence, toxoplasmosis has received relatively little attention compared to other infectious diseases. Wilson hopes her team’s work will help shift that perspective.
“Our work changes how we think about the Toxoplasma cyst,” she said. “It reframes the cyst as the central control point of the parasite’s life cycle. It shows us where to aim new treatments. If we want to really treat toxoplasmosis, the cyst is the place to focus.”
Wilson was joined in the study by Arzu Ulu, Sandeep Srivastava, Nala Kachour, Brandon H. Le, and Michael W. White. Wilson and White are co-corresponding authors of the paper.
The study was supported by grants from the National Institute of Allergy and Infectious Diseases of the National Institutes of Health.
The title of the paper is “Bradyzoite subtypes rule the crossroads of Toxoplasma development.”




