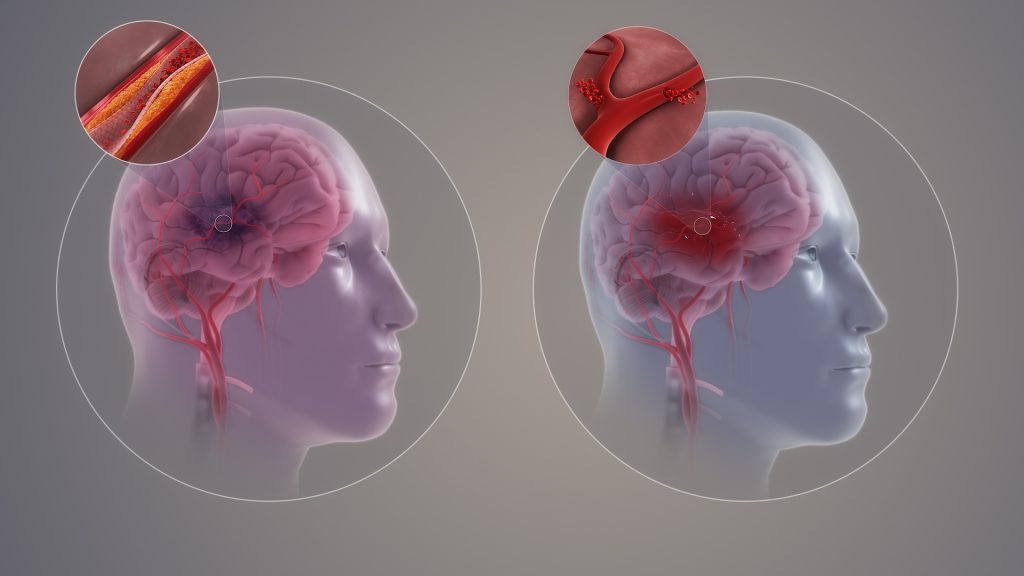
COVID Infection Linked to MI & Stroke Risk Increases up to 3 Years Later
An analysis of data in the UK Biobank has found that COVID infection may increase the risk of myocardial infarction (MI), stroke and death from any cause for up to three years for people with and without cardiovascular disease, according to new research published in the American Heart Association’s peer-reviewed journal Arteriosclerosis, Thrombosis and Vascular Biology (ATVB).
“We found a long-term cardiovascular health risk associated with COVID, especially among people with more severe COVID cases that required hospitalisation,” said lead study author James Hilser, M.P.H., Ph.D.-candidate at the University of Southern California Keck School of Medicine in Los Angeles. “This increased risk of heart attack and stroke continued three years after COVID infection. Remarkably, in some cases, the increased risk was almost as high as having a known cardiovascular risk factor such as Type 2 diabetes or peripheral artery disease.”
Previous research has shown that COVID increases the risk of serious cardiovascular complications within the first month after infection. This study examined how long the increased risk lasted and whether it subsided after recovering from COVID infection.
Researchers reviewed health and genetic data in the UK Biobank for more than 10 000 adults, including approximately 8000 who had tested positive for SARS-CoV-2 from February 1 to December 31, 2020 and about 2000 who tested positive for the virus in a hospital setting in 2020. A group of more than 200,000 adults who had no history of COVID infection during the same time frame in the UK Biobank were also reviewed for comparison. None of the participants were vaccinated at the time of infection because COVID vaccines were not yet available in 2020.
The analysis found:
- During the nearly 3-year follow-up period, the risk of heart attack, stroke and death was more than two times higher among adults who had COVID, and nearly four times greater among adults hospitalized with COVID, compared with the group with no history of COVID infection.
- People hospitalized with COVID, without cardiovascular disease or without Type 2 diabetes, had a 21% greater risk of heart attack, stroke and death compared to people with cardiovascular disease and without COVID infection.
- There was a significant genetic interaction among the non-O blood types and hospitalisation for COVID. People with severe COVID infections had an increased risk of heart attack and stroke, however, that risk was even higher in people who had non-O blood types (those with blood types A, B or AB).
- The risk of heart attack and stroke was about 65% higher in adults with non-O blood types compared to those who had type O blood. A preliminary analysis did not show that Rh (positive or negative) blood type interacted with severe COVID, the authors noted.
“Worldwide, over a billion people have already experienced COVID infection. The findings reported are not a small effect in a small subgroup,” said co-senior study author Stanley Hazen, M.D., Ph.D., chair of cardiovascular and metabolic sciences in Cleveland Clinic’s Lerner Research Institute and co-section head of preventive cardiology. “The results included nearly a quarter million people and point to a finding of global health care importance that may translate into an explanation for a rise in cardiovascular disease around the world.”
Study details, background and design:
- Health data was from the UK Biobank, a large-scale study of 503,325 adults living in the United Kingdom who were 40 to 69 years of age at enrollment between 2006 and 2010. The in-depth health and biomedical information was collected for participants registered in the UK National Health Service with a UK general practitioner (similar to a primary care physician in the U.S.).
- This analysis included health data for 10,005 adults who tested positive for the COVID virus or were hospitalized with COVID between February 1, 2020, and December 31, 2020. An additional 217,730 peers enrolled in the UK Biobank who did not have COVID during the same time period were included. In the analysis, all participants were matched as closely as possible for demographics and similar health conditions.
- Major adverse cardiovascular events (heart attack, stroke and all-cause death) were evaluated for long-term risk, through October 31, 2022, approximately 3 years later.
“This interesting paper is really two studies in one,” said Sandeep R. Das, M.D., M.P.H., MBA, FAHA, co-chair of the American Heart Association’s COVID-19 CVD Registry committee and director for quality and value in the cardiology division for UT Southwestern Medical Center in Dallas. “First, the authors show that having been hospitalized with COVID is a marker of increased cardiovascular risk, on par with having a pre-existing diagnosis of cardiovascular disease. Although proving direct cause and effect is very difficult to tease out in a study that only analyses past data collected for other purposes, this finding is important because it suggests a history of prior COVID hospitalization, even without a history of CVD, should be considered to initiate and possibly accelerate CVD prevention efforts. Whether severe COVID infection has a direct impact on the vascular system is an interesting area for study as well,” Das said.
“The second ‘study’ in this paper looks at the relationship between ABO blood type and COVID outcomes. They show that something located close to the genetic home of ABO blood type is associated with different degrees of susceptibility to COVID. This is really fascinating, and I look forward to seeing scientists tease out what the specific pathway may be.”
The study had several limitations, including that the data was from patients who had the original strain of the COVID virus before vaccines were widely available in 2021. Additionally, the researchers noted that UK Biobank information on medication use was not specific to the beginning of the pandemic in 2020 or the date that patients were infected with SARS-CoV-2. Also, because the majority of participants in the UK Biobank are white, additional research is needed to confirm that these results apply to people with diverse racial and ethnic backgrounds.
“The results of our study highlight the long-term cardiovascular effects of COVID infection. Given the increased risk of heart attack, stroke and death, the question is whether or not severe COVID should be considered as another risk factor for CVD, much like Type 2 diabetes or peripheral artery disease, where treatment focused on CVD prevention may be valuable,” said co-senior study author Hooman Allayee, Ph.D., a professor of population and public health sciences at the University of Southern California Keck School of Medicine in Los Angeles. “The results suggest that people with prior COVID infection may benefit from preventive care for cardiovascular disease.”
Source: American Heart Association