Fibroblasts Have Hidden Powers That Could Heal Brain Injuries
Healing from any injury involves a delicate balance between scarring and inflammation – two processes that can wreak havoc as well as make repairs.
When the injury is to the brain, the balance is that much more important, yet scientists know almost nothing of how this process works.
Now, a study from UC San Francisco spotlights how a cell type called a fibroblast, that plays a healing role in other parts of the body also performs a similar function in the brain. The discovery is a step toward finding new ways to treat brain injuries, which are the nation’s leading cause of death and disability and for which there aren’t any drugs that can intervene.
Fibroblasts were only identified in the brain in the last decade. They reside mostly in the meninges, a set of protective membranes that surround the brain and spinal cord. Until now, scientists thought they mostly served to maintain the structure of the meninges and its network of blood vessels.
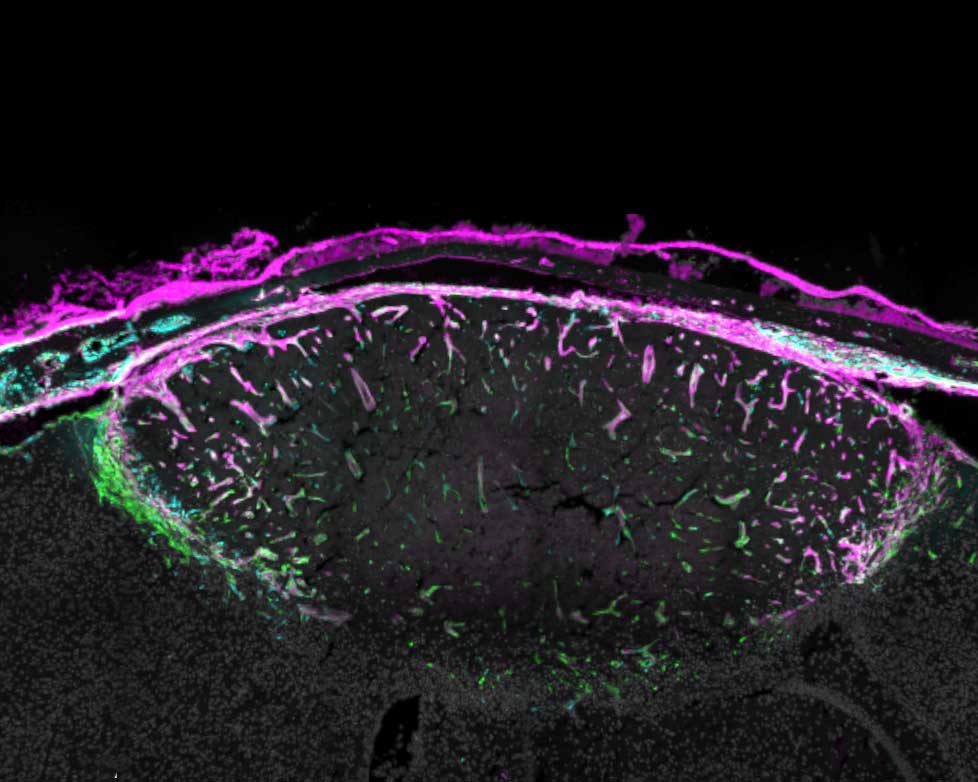
Ari Molofsky, MD, PhD, a professor of laboratory medicine, suspected the fibroblasts might be doing much more than that. He and Tom Arnold, MD, a professor of paediatrics, discovered that when the brain is injured – whether from a blow or a stroke – fibroblasts navigate from the meninges and surround the injured tissue where they create a protective barrier, or scar.
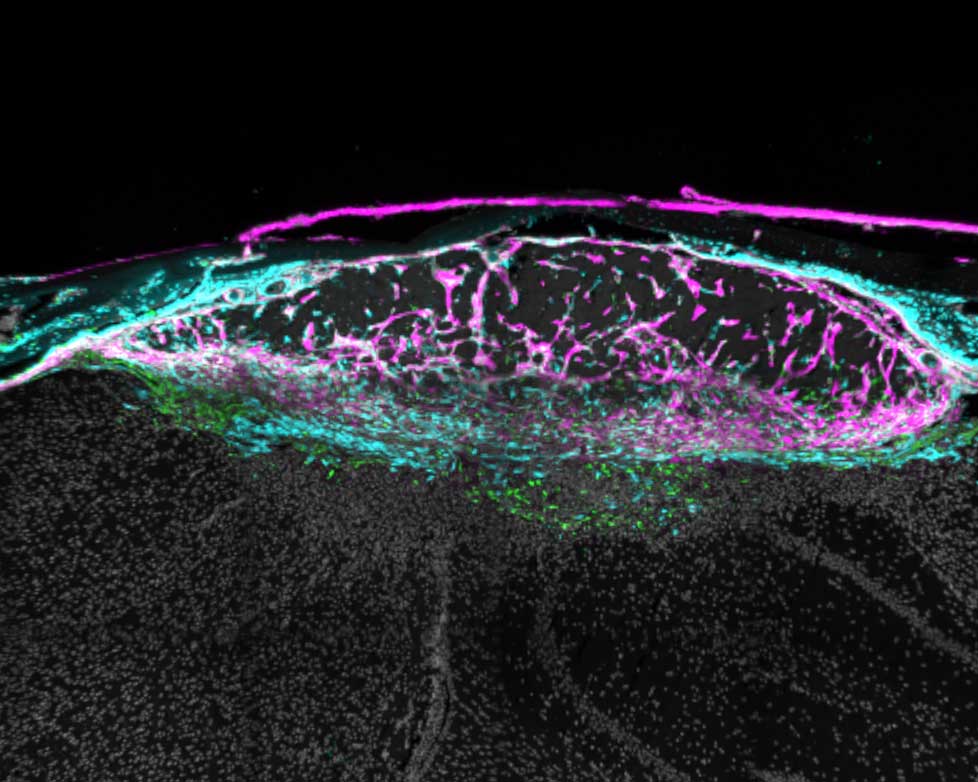
Then, about a week later, after the scar has formed, the fibroblasts adopt new roles. Some recruit immune cells that are required for healing; others ensure that the immune response doesn’t cause too much inflammation; and still others return to the meninges. Understanding these distinct stages could spur new interventions to help people with serious injuries.
“Our study reveals opportunities to enhance the natural repair process,” said Molofsky, the senior author of the study, which appeared in Nature. “The goal is to give someone who’s experienced a traumatic brain injury or stroke the best outcome possible, based on the stage of healing they’re in.”
Therapies currently in clinical trials for lung and liver fibrosis target a molecule that prompts fibroblasts to create scarring. This suggests that other similar drugs could enhance healing in the early stages of a brain injury.
Molofsky’s study also offers an ideal venue for scientists to learn how fibroblasts are doing their work elsewhere in the body. Being largely devoid of immune cells, the brain offers a much clearer view than other organs like the lungs or liver, where immune cells may be too crowded around fibroblasts to see what they are doing.
“There’s a lot of potential here,” Molofsky said. “These overlooked cells seem adept at solving the common challenge of balancing healing and inflammation.”