The Thymus May Be Critical for Longevity and Immunotherapy
Mass General Brigham researchers used artificial intelligence to analyse routine medical scans, uncovering how the thymus impacts aging, cardiovascular risk, cancer incidence, and response to immunotherapy.
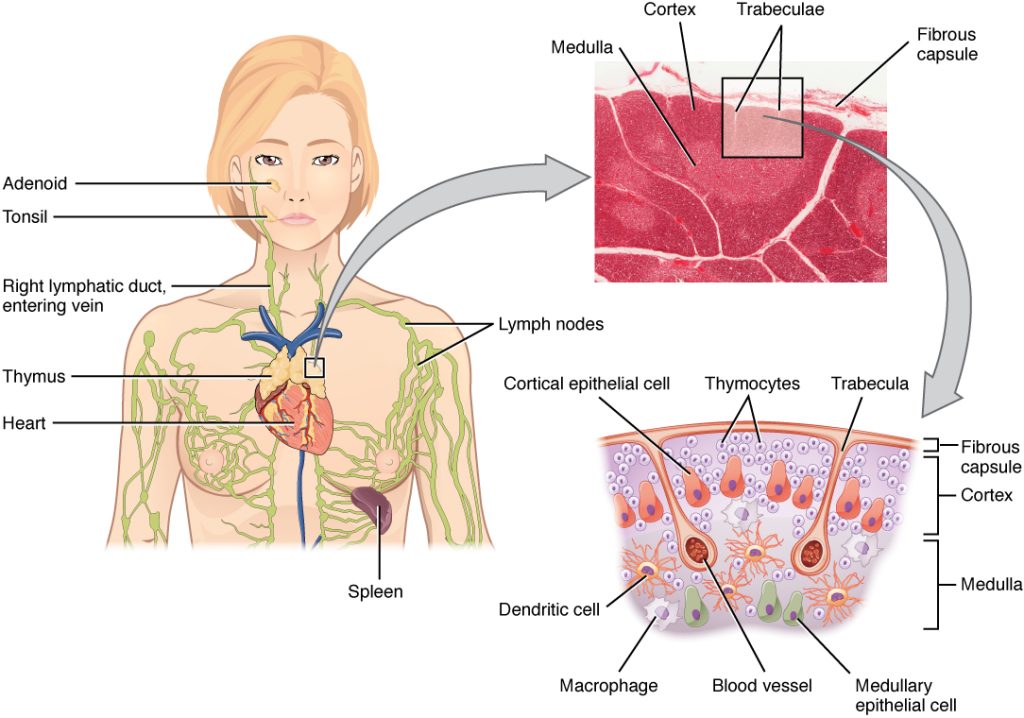
Two new studies from investigators at Mass General Brigham challenge a decades-old assumption that the thymus, an organ best known for its role in establishing immune function in childhood, becomes irrelevant in adulthood. Using artificial intelligence (AI) to analyse routine CT scans, researchers uncovered that adults with a healthy thymus had increased longevity and reduced risk for cardiovascular disease and cancer. In a separate study of patients with cancer, the researchers found that thymic health may influence response to immunotherapy – a treatment that depends on the strength of a patient’s immune system.
These findings, published in two papers in the same issue of Nature, suggest the thymus plays a far more consequential role in adult health than previously understood, and might provide a new target for personalising disease prevention and cancer treatments.
“The thymus has been overlooked for decades and may be a missing piece in explaining why people age differently, and why cancer treatments fail in some patients,” said Hugo Aerts, PhD, corresponding author on the papers and director of the Artificial Intelligence in Medicine (AIM) Program at Mass General Brigham. “Our findings suggest thymic health deserves much more attention and may open new avenues for understanding how to protect the immune system as we age.”
The thymus is a small organ in the chest that helps train T cells, priming the immune system to protect the body from infections and disease. For decades, doctors believed the organ was mostly inactive after puberty because it shrinks with age and produces fewer new T cells. As a result, its role in adult health has rarely been examined in large populations.
Previous research has linked T cell diversity to aging and immune decline, but most of those studies relied on small, blood-based analyses. In contrast, the new studies examined more than 25 000 adults in a national lung cancer screening trial and over 2500 participants in the Framingham Heart Study – a large, long-running population cohort of generally healthy adults.
The team analysed the size, shape, and composition of the thymus, generating a “thymic health” score. People with high thymic health scores had about a 50% lower risk of death, 63% lower risk of cardiovascular death, and 36% lower risk of developing lung cancer compared to those with low thymic health. These associations remained significant after adjusting for age and other health factors.
The researchers theorize that when thymic health and T cell diversity decline, the immune system may become less able to respond to new threats, like cancer or other diseases. Their analysis found that chronic inflammation, smoking, and high body weight were associated with poorer thymic health, suggesting that lifestyle and systemic inflammation may influence immune resilience across the lifespan.
In a second study, the researchers analysed CT scans and outcomes from more than 1200 immunotherapy-treated patients. Patients with stronger thymic health had about a 37% lower risk of cancer progression and a 44% lower risk of death, even after accounting for other patient, tumour, and treatment factors. These findings point to a previously underappreciated role the thymus may play in shaping how well patients respond to modern cancer immunotherapies.
The researchers caution that their findings will need to be confirmed in future studies, and the imaging method is not yet ready for routine clinical use. While lifestyle factors were linked to thymic health, the studies did not test whether modifying those factors can directly improve thymic function.
The team is currently leading additional research to investigate whether other care-associated factors may impact thymic health. In one study, they are examining whether unintended radiation exposure to the thymus in patients with lung cancer may affect outcomes.
“Improving our understanding and monitoring of thymic health could eventually help physicians better assess disease risk and guide treatment decisions,” said Aerts.
Source: Mass General Brigham