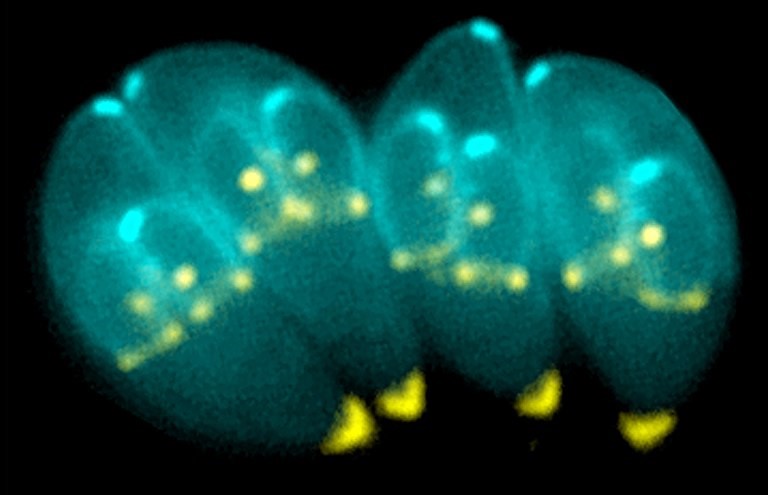
Credit: Image provided by Ke Hu and John M. Murray
A new method blocks the protein regulation of Toxoplasma gondii, causing the parasite to die off inside the host cell, a method which could be adapted to malaria. The approach is detailed in the journal Nature Microbiology.
Toxoplasmosis is one of the most widespread zoonoses worldwide. It is an infectious disease that can be transmitted from cats to humans, and from consumption of raw or undercooked meat. Infection is particularly dangerous for pregnant women, and immunosuppressed HIV/AIDS patients often manifest neurological symptoms.
The cause of the disease is the single-celled parasite T. gondii. Inside the host cell, it forms a parasitophorous vacuole, a tiny compartment facilitating nutrient exchange and synchronised cell division. Up to 64 daughter cells can form inside, connected with each other inside the vacuole via a network. As soon as the offspring are mature, a regulation mechanism prompts the dissolution of the vacuole and the structures that have formed inside it, releasing the daughter cells to invade new host cells.
Hope for the development of new drugs
Previously, it was not known which genes encode the proteins that control the exit from the host cell. To identify them, a team led by Professor Markus Meißner at LMU, collaborated with colleagues from the University of Glasgow in Scotland to develop a novel genetic screening technique based on Cas9 ‘genetic scissors’, and investigate a library of 320 parasite-specific genes. They discovered two genes without which cell egress is impossible.
The targeted destruction of these genes resulted in the exit being trapped and the next generation of parasites dying within the host cell. “This paves the way potentially for the development of active substances that could block the function of the corresponding proteins and so put a halt to propagation,” remarked Prof Markus Meißner.
T. gondii is closely related to the malaria pathogen Plasmodium falciparum. Therefore, the parasite serves as a model organism for the pathogen of the tropical disease, which kills hundreds of thousands of people worldwide every year. “We assume that similar processes control the propagation of the malaria pathogen,” explains LMU parasitologist Dr. Elena Jimenez-Ruiz. “Next, we will investigate what functions these proteins have in the malaria pathogen and whether there are possible starting points for the development of new drugs.”

