Extended Chemotherapy Slashes Childhood Leukaemia Recurrence
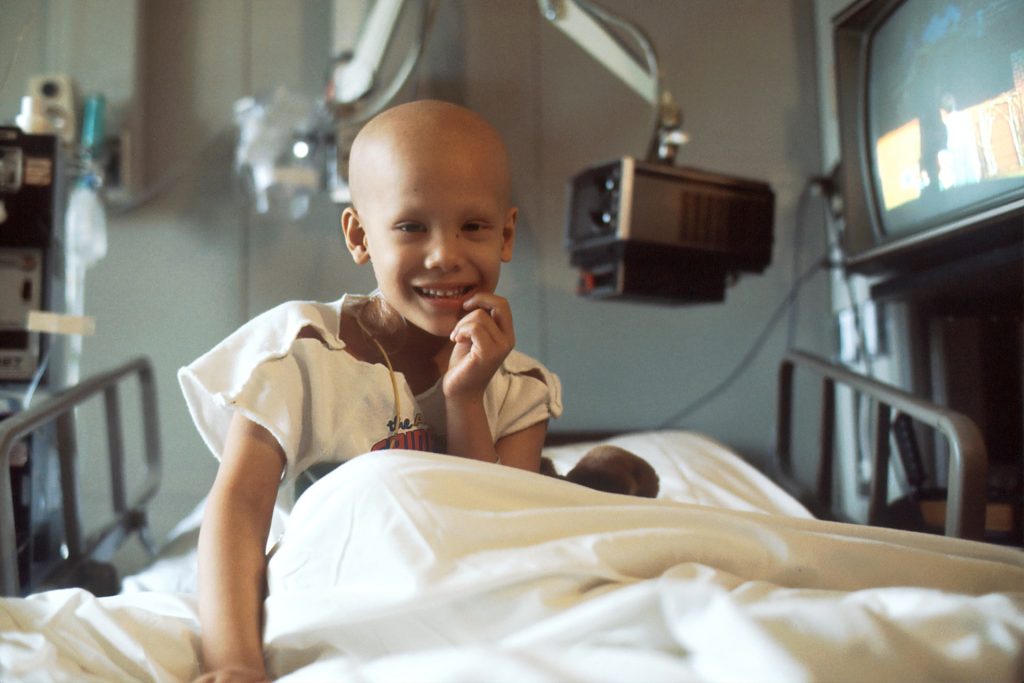
Giving three years of chemotherapy to children with acute lymphoblastic leukaemia (ALL) instead of two years lowers the risk of their disease coming back after treatment by three times. The survival rate of all children with ALL, the most common form of childhood cancer, together has further increased to 94%. Less intensive therapy proved safe for three groups of children, resulting in a better quality of life. These findings on a large Dutch study into ALL were reported at the annual conference of the American Society of Hematology (ASH).
Many children with ALL have good outcomes. After two years of chemotherapy treatment, nine out of ten children are cured. But some children have a more aggressive disease, such as having the Ikaros mutation in their leukaemia cells, have a greater risk of recurrence after treatment. In order to improve the chances of survival and quality of life of all children with leukaemia, the treatment protocol has been continuously adapted over the years, based on the latest scientific insights.
Prof Rob Pieters, medical director and paediatric oncologist at the Princess Máxima Center for paediatric oncology in the Netherlands, presented the outcomes of the ALL-11 treatment protocol. The Dutch researchers tested the benefit of an adapted treatment in specific groups of children with leukaemia, including children with the Ikaros mutation. More than 800 children in the Netherlands were treated with this protocol between April 2012 and July 2020.
Threefold lower risk of recurrence
Children with Ikaros leukaemia received an extra year of chemotherapy in the ‘maintenance phase’ on top of the first two years of treatment. This change lowered the risk of their cancer coming back by threefold: this happened in only 9% of them, compared to 26% of the children in the previous treatment protocol.
87% of children with Ikaros leukaemia survived their disease for five years without their cancer coming back, an improvement on the 72% in the previous protocol. Because of the extra year of chemotherapy, this group of children had a slightly higher risk of infection, but these were treatable. The extended therapy did not lead to any additional side effects.
Analysis of data from all children with ALL, regardless of subtype, showed that the five-year survival rate has improved stepwise over the past 30 years from 80% to 94% under the ALL-11 protocol.
Safe reduction of treatment
In the ALL-11 protocol, doctors and researchers also looked at the benefit of a less intensive treatment plan for three groups of children. This included children with a leukaemia mutation linked to a very high chance of recovery, and children with Down syndrome who experience more severe side effects. These children received treatment without or with a lower dose of anthracyclines, a type of leukaemia drug that increases the risk of heart damage and infections. The reduced treatment proved successful: children had the same or even a better chance of survival, while their quality of life improved due to a lower risk of infections and damage to the heart.
Global interest
Globally, there is much interest in the Dutch research as it has been unclear how to improve therapy for children with Ikaros leukaemia. The results have now been presented for the first time at the largest blood cancer conference, and could lead to changes in treatment protocols for these children worldwide.
In the Netherlands, there are about 15 children with ALL each year for whom existing treatments stop working. Since 2019, they have been eligible for treatment with CAR T-cell therapy, a promising form of immunotherapy that now leads to a cure in 40% of these children.
Making a difference
Prof Monique den Boer, medical biologist and group leader at the Princess Máxima Center, played an important role in the adapted therapy for children with the Ikaros gene change. She says: ‘The Ikaros mutation was first discovered about 15 years ago in children with leukaemia who had a poor prognosis, partly thanks to the emergence of new DNA technologies. We saw that the cancer came back in many of these children shortly after the end of the two-year treatment plan. I am very proud that our lab findings have now found their way into the clinic and can make such a big difference for children with leukaemia.”
More cure with fewer side effects
Prof Pieters concludes: The five-year survival rate for children with acute lymphoblastic leukaemia has increased enormously since the 1960s, from zero to 94%, but the last steps are the most difficult. We are now one step closer to curing all children with ALL. We have also largely been able to remove a drug that poses a risk of heart damage from the treatment of children with a less aggressive form of the disease. The latest results for children with leukaemia therefore fit in perfectly with our mission: curing more children with cancer, with fewer side effects.”
Source: Princess Máxima Center